|
|
| (4 intermediate revisions by one other user not shown) |
| Line 21: |
Line 21: |
| | height: 100%; | | height: 100%; |
| | z-index: 9999; | | z-index: 9999; |
| − | background: #2B2B35; | + | background: #292935; |
| | } | | } |
| | | | |
| Line 67: |
Line 67: |
| | <body> | | <body> |
| | <div class="se-pre-con"> | | <div class="se-pre-con"> |
| − | <img src="https://static.igem.org/mediawiki/2019/8/83/T--Tuebingen--preloader.gif"> | + | <img src="https://static.igem.org/mediawiki/2019/4/49/T--Tuebingen--DNA_gif.gif"> |
| | </div> | | </div> |
| | <script src="https://2019.igem.org/Team:Tuebingen/jqueryLoadJS?action=raw&ctype=text/javascript"></script> | | <script src="https://2019.igem.org/Team:Tuebingen/jqueryLoadJS?action=raw&ctype=text/javascript"></script> |
| Line 9,131: |
Line 9,131: |
| | margin-bottom: 0px; | | margin-bottom: 0px; |
| | } | | } |
| − |
| |
| | | | |
| | | | |
| Line 10,236: |
Line 10,235: |
| | <body style=""> | | <body style=""> |
| | <section id="nav-placeholder"> | | <section id="nav-placeholder"> |
| − | <nav class="shadow navbar navbar-expand-md navbar-dark fixed-top"
| + | <nav class="shadow navbar navbar-expand-md navbar-dark fixed-top" |
| | style="background-color: rgba(30, 30, 30, 0.3); z-index: 100;"> | | style="background-color: rgba(30, 30, 30, 0.3); z-index: 100;"> |
| | <a class="navbar-brand" href="https://2019.igem.org/Team:Tuebingen"> | | <a class="navbar-brand" href="https://2019.igem.org/Team:Tuebingen"> |
| Line 10,263: |
Line 10,262: |
| | style="font-family: 'Righteous', cursive; font-weight: normal; color: #2ecc71;">Description</a> | | style="font-family: 'Righteous', cursive; font-weight: normal; color: #2ecc71;">Description</a> |
| | <a class="dropdown-item" href="/Team:Tuebingen/Killswitch" | | <a class="dropdown-item" href="/Team:Tuebingen/Killswitch" |
| − | style="font-family: 'Righteous', cursive; font-weight: normal; color: #2ecc71;">Kill Switch</a> | + | style="font-family: 'Righteous', cursive; font-weight: normal; color: #2ecc71;">Kill |
| | + | Switch</a> |
| | <a class="dropdown-item" href="/Team:Tuebingen/Nissle" | | <a class="dropdown-item" href="/Team:Tuebingen/Nissle" |
| | style="font-family: 'Righteous', cursive; font-weight: normal; color: #2ecc71;">Nissle</a> | | style="font-family: 'Righteous', cursive; font-weight: normal; color: #2ecc71;">Nissle</a> |
| Line 10,341: |
Line 10,341: |
| | style="font-family: 'Righteous', cursive; font-weight: normal; color: #2ecc71;">Overview</a> | | style="font-family: 'Righteous', cursive; font-weight: normal; color: #2ecc71;">Overview</a> |
| | <a class="dropdown-item" href="/Team:Tuebingen/Public_Engagement" | | <a class="dropdown-item" href="/Team:Tuebingen/Public_Engagement" |
| − | style="font-family: 'Righteous', cursive; font-weight: normal; color: #2ecc71;">Education & Public Engagement</a> | + | style="font-family: 'Righteous', cursive; font-weight: normal; color: #2ecc71;">Education & |
| | + | Public Engagement</a> |
| | <a class="dropdown-item" href="/Team:Tuebingen/Experts" | | <a class="dropdown-item" href="/Team:Tuebingen/Experts" |
| | style="font-family: 'Righteous', cursive; font-weight: normal; color: #2ecc71;">Experts</a> | | style="font-family: 'Righteous', cursive; font-weight: normal; color: #2ecc71;">Experts</a> |
| | <a class="dropdown-item" href="/Team:Tuebingen/SDG" | | <a class="dropdown-item" href="/Team:Tuebingen/SDG" |
| − | style="font-family: 'Righteous', cursive; font-weight: normal; color: #2ecc71;">Sustainable Development Goals</a> | + | style="font-family: 'Righteous', cursive; font-weight: normal; color: #2ecc71;">Sustainable |
| | + | Development Goals</a> |
| | <a class="dropdown-item" href="/Team:Tuebingen/Experimenta" | | <a class="dropdown-item" href="/Team:Tuebingen/Experimenta" |
| | style="font-family: 'Righteous', cursive; font-weight: normal; color: #2ecc71;">Experimenta | | style="font-family: 'Righteous', cursive; font-weight: normal; color: #2ecc71;">Experimenta |
| Line 10,360: |
Line 10,362: |
| | <div class="dropdown-menu"> | | <div class="dropdown-menu"> |
| | <a class="dropdown-item" href="/Team:Tuebingen/Human_Practices" | | <a class="dropdown-item" href="/Team:Tuebingen/Human_Practices" |
| − | style="font-family: 'Righteous', cursive; font-weight: normal; color: #2ecc71;">Integrated Human Practise</a> | + | style="font-family: 'Righteous', cursive; font-weight: normal; color: #2ecc71;">Integrated |
| | + | Human Practise</a> |
| | <a class="dropdown-item" href="/Team:Tuebingen/Public_Engagement" | | <a class="dropdown-item" href="/Team:Tuebingen/Public_Engagement" |
| − | style="font-family: 'Righteous', cursive; font-weight: normal; color: #2ecc71;">Education & Public Engagement</a> | + | style="font-family: 'Righteous', cursive; font-weight: normal; color: #2ecc71;">Education & |
| | + | Public Engagement</a> |
| | <a class="dropdown-item" href="/Team:Tuebingen/Entrepreneurship" | | <a class="dropdown-item" href="/Team:Tuebingen/Entrepreneurship" |
| − | style="font-family: 'Righteous', cursive; font-weight: normal; color: #2ecc71;">Supporting Entrepreneurship</a> | + | style="font-family: 'Righteous', cursive; font-weight: normal; color: #2ecc71;">Supporting |
| | + | Entrepreneurship</a> |
| | <a class="dropdown-item" href="/Team:Tuebingen/Model" | | <a class="dropdown-item" href="/Team:Tuebingen/Model" |
| | style="font-family: 'Righteous', cursive; font-weight: normal; color: #2ecc71;">Model</a> | | style="font-family: 'Righteous', cursive; font-weight: normal; color: #2ecc71;">Model</a> |
| Line 10,379: |
Line 10,384: |
| | <li class="nav-item mx-3"> | | <li class="nav-item mx-3"> |
| | <a class="nav-link" href="https://igem.org/2019_Judging_Form?team=Tuebingen" target="_blank" | | <a class="nav-link" href="https://igem.org/2019_Judging_Form?team=Tuebingen" target="_blank" |
| − | style="font-family: 'Righteous', cursive; font-weight: normal; color: #2ecc71;">JUDGING FORM ⇗</a> | + | style="font-family: 'Righteous', cursive; font-weight: normal; color: #2ecc71;">JUDGING FORM |
| | + | ⇗</a> |
| | </li> | | </li> |
| | | | |
| Line 10,471: |
Line 10,477: |
| | opacity: 1; | | opacity: 1; |
| | text-decoration: none; | | text-decoration: none; |
| − | color: rgba(105, 202, 98, 1.1); | + | color: #2ecc71; |
| | } | | } |
| | | | |
| Line 10,477: |
Line 10,483: |
| | opacity: 1; | | opacity: 1; |
| | text-decoration: none; | | text-decoration: none; |
| − | color: rgba(105, 202, 98, 1.1); | + | color: #2ecc71; |
| | } | | } |
| | | | |
| Line 10,501: |
Line 10,507: |
| | */ | | */ |
| | .btn-one { | | .btn-one { |
| − | color: rgba(105, 202, 98, 1.1); | + | color: #2ecc71; |
| | transition: all 0.3s; | | transition: all 0.3s; |
| | position: relative; | | position: relative; |
| Line 10,524: |
Line 10,530: |
| | border-top-style: solid; | | border-top-style: solid; |
| | border-bottom-style: solid; | | border-bottom-style: solid; |
| − | border-top-color: rgba(105, 202, 98, 1.1); | + | border-top-color: #2ecc71; |
| − | border-bottom-color: rgba(105, 202, 98, 1.1); | + | border-bottom-color: #2ecc71; |
| | transform: scale(0.1, 1); | | transform: scale(0.1, 1); |
| | } | | } |
| Line 10,761: |
Line 10,767: |
| | <div class="card-body" style="background-color: #232323;"> | | <div class="card-body" style="background-color: #232323;"> |
| | <p style="color: #F4F0DE"> | | <p style="color: #F4F0DE"> |
| − | <b>08 Jakob Patrick Eva</b> After moving into our new lab and settling in, first practical | + | <b>08 Jakob Patrick Eva</b> After moving into our new lab and settling in, first |
| | + | practical |
| | steps like aliquoting our primers or preparing LB-Medium and LB-Agar were taken. | | steps like aliquoting our primers or preparing LB-Medium and LB-Agar were taken. |
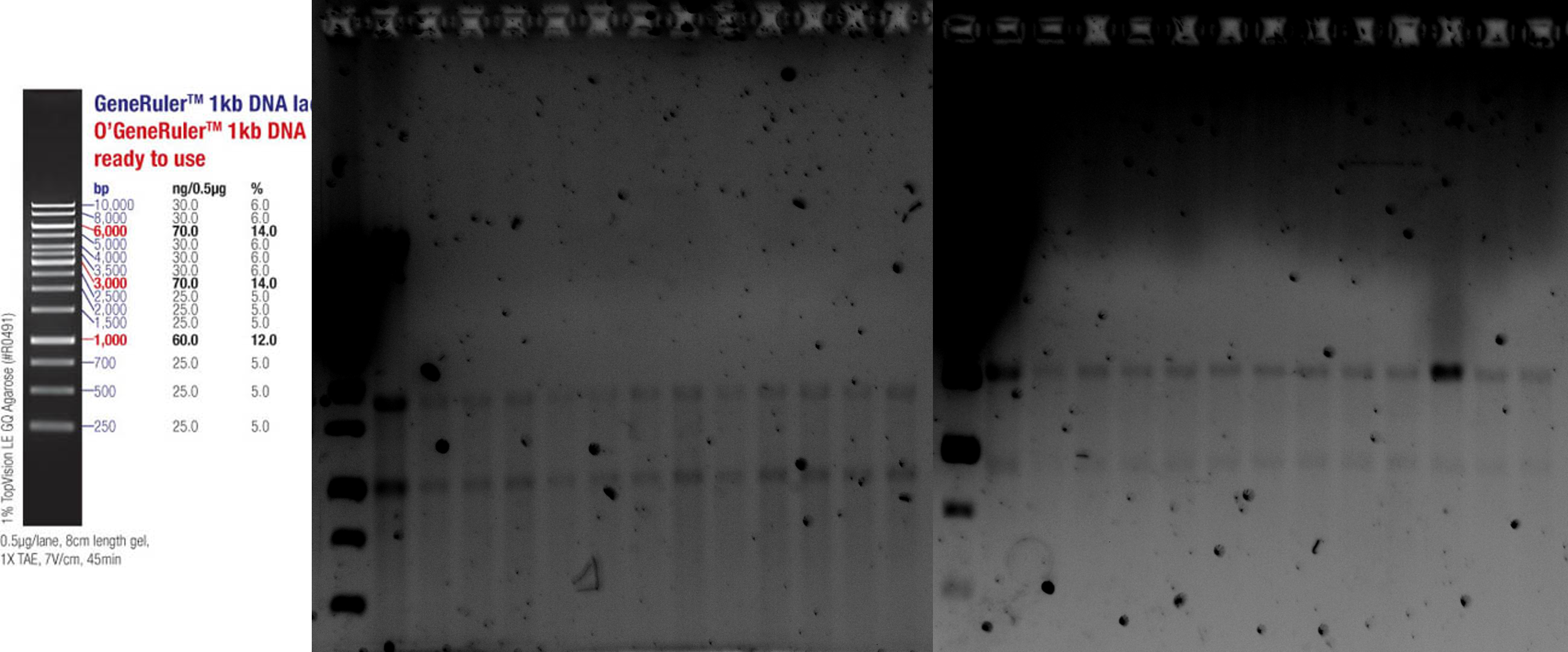
| − | <b>09 Jakob Patrick Eva</b> We amplified the cas3 gene out of <i>E. coli</i> gDNA by PCR | + | <b>09 Jakob Patrick Eva</b> We amplified the cas3 gene out of <i>E. coli</i> |
| − | (Q5 Polymerase) and transformed the biobricks BBa_K608351 (K2), BBa_K091001 (K5), | + | gDNA by PCR |
| | + | (Q5 Polymerase) and transformed the biobricks BBa_K608351 (K2), BBa_K091001 |
| | + | (K5), |
| | BBa_I13453 (K10), BBa_K584000 (AraC Promotor), BBa_K117008 (LsrR Promotor), | | BBa_I13453 (K10), BBa_K584000 (AraC Promotor), BBa_K117008 (LsrR Promotor), |
| | BBa_R0073 (Mnt Promotor) in competent <i>E. coli</i> DH5-Alpha (NEB iGEM Kit). | | BBa_R0073 (Mnt Promotor) in competent <i>E. coli</i> DH5-Alpha (NEB iGEM Kit). |
| − | <b>10 Eva Patrick</b> We amplified cascade genes out of <i>E. coli</i> MG1655 gDNA via PCR | + | <b>10 Eva Patrick</b> We amplified cascade genes out of <i>E. coli</i> MG1655 |
| | + | gDNA via PCR |
| | (Q5 Polymerase) and checked the amplification of cas3 and cascade via agarose | | (Q5 Polymerase) and checked the amplification of cas3 and cascade via agarose |
| | gel electrophoresis. (Fig. 1) We picked colonies for all but the Ba_K608351 | | gel electrophoresis. (Fig. 1) We picked colonies for all but the Ba_K608351 |
| | (K2) transformations (no growth) for inoculating overnight cultures and made | | (K2) transformations (no growth) for inoculating overnight cultures and made |
| − | a new batch of competent <i>E. coli</i> DH5-Alpha cells. <b>11 Jakob Eva Patrick</b> | + | a new batch of competent <i>E. coli</i> DH5-Alpha cells. <b>11 Jakob Eva |
| | + | Patrick</b> |
| | By Miniprep (Qiagen) we extracted the DNA of the overnight cultures. | | By Miniprep (Qiagen) we extracted the DNA of the overnight cultures. |
| | Amplification via PCR (Q5 Polymerase) of cascade was repeated (Fig. 2). | | Amplification via PCR (Q5 Polymerase) of cascade was repeated (Fig. 2). |
| Line 10,785: |
Line 10,796: |
| | class="figure-img img-fluid rounded" | | class="figure-img img-fluid rounded" |
| | alt="A generic square placeholder"></a> | | alt="A generic square placeholder"></a> |
| − | <figcaption style="color: #F4F0DE; font-size: small;">Fig. 1. 17/10/19 Gel elctrophoresis of cas3 and cascade | + | <figcaption style="color: #F4F0DE; font-size: small;">Figure 1: 17/10/19 Gel |
| | + | elctrophoresis of cas3 and cascade |
| | PCR. Gel loaded (from left to right): Ladder 1kb, cas3 (72°C), cas3 (68°C), | | PCR. Gel loaded (from left to right): Ladder 1kb, cas3 (72°C), cas3 (68°C), |
| − | H2O, cascade (72°C), cascade (68°C), H2O. It shows positive PCR of cas3 (68°C), expected at ~2.7 kb. | + | H2O, cascade (72°C), cascade (68°C), H2O. It shows positive PCR of cas3 |
| | + | (68°C), expected at ~2.7 kb. |
| | </figcaption> | | </figcaption> |
| | </figure> | | </figure> |
| Line 10,798: |
Line 10,811: |
| | class="figure-img img-fluid rounded" | | class="figure-img img-fluid rounded" |
| | alt="A generic square placeholder image"></a> | | alt="A generic square placeholder image"></a> |
| − | <figcaption style="color: #F4F0DE; font-size: small;">Fig. 2. 07/11/19 Gel electrophoresis of cascade PCR. | + | <figcaption style="color: #F4F0DE; font-size: small;">Figure 2: 07/11/19 Gel |
| | + | electrophoresis of cascade PCR. |
| | Gel loaded (from left to right): Ladder 1kb, cascade (68°C), cascade (64°C). | | Gel loaded (from left to right): Ladder 1kb, cascade (68°C), cascade (64°C). |
| | It shows positive PCR of cas3 (68°C), expected at ~4.4 kb. | | It shows positive PCR of cas3 (68°C), expected at ~4.4 kb. |
| Line 10,822: |
Line 10,836: |
| | <div class="card-body" style="background-color: #232323;"> | | <div class="card-body" style="background-color: #232323;"> |
| | <p style="color: #F4F0DE"> | | <p style="color: #F4F0DE"> |
| − | <b>15 Jakob Patrick Eva Luzi</b> Transformations of DH5-Alpha cells with the biobricks | + | <b>15 Jakob Patrick Eva Luzi</b> Transformations of DH5-Alpha cells with the |
| | + | biobricks |
| | were repeated. 16 Patrick, Eva Since there repeatedly weren’t any colonies | | were repeated. 16 Patrick, Eva Since there repeatedly weren’t any colonies |
| | for BBa_K608351 (K2), we transformed NovaBlue™ competent cells with this | | for BBa_K608351 (K2), we transformed NovaBlue™ competent cells with this |
| | biobrick. Overnight cultures of the other biobricks as well as an overnight | | biobrick. Overnight cultures of the other biobricks as well as an overnight |
| − | culture of <i>E. coli</i> Nissle were inoculated. <b>17 Eva Patrick Katharina</b> Glycerol | + | culture of <i>E. coli</i> Nissle were inoculated. <b>17 Eva Patrick |
| − | stocks of the overnight cultures were prepared. Competent <i>E. coli</i> Nissle cells | + | Katharina</b> Glycerol |
| − | were produced and the growth of <i>E. coli</i> Nissle was analysed preliminarily. | + | stocks of the overnight cultures were prepared. Competent <i>E. coli</i> Nissle |
| | + | cells |
| | + | were produced and the growth of <i>E. coli</i> Nissle was analysed |
| | + | preliminarily. |
| | After Miniprep (Qiagen) of BBa_K516030 (rfp) a PCR (Pfu Polymerase) was run. | | After Miniprep (Qiagen) of BBa_K516030 (rfp) a PCR (Pfu Polymerase) was run. |
| | After digestion of the PCR products cas3 and cascade as well as the plasmid | | After digestion of the PCR products cas3 and cascade as well as the plasmid |
| − | BBa_I13453 (K10), K10_cascade and K10_cas3 were ligated. <b>18 Eva Patrick</b> The | + | BBa_I13453 (K10), K10_cascade and K10_cas3 were ligated. <b>18 Eva Patrick</b> |
| | + | The |
| | K10_cascade and K10_cas3 ligations were digested (single) and analysed by gel | | K10_cascade and K10_cas3 ligations were digested (single) and analysed by gel |
| | electrophoresis, together with the rfp PCR product (Fig. 3). The gel did not | | electrophoresis, together with the rfp PCR product (Fig. 3). The gel did not |
| | show a result for K10_cascade and K10_cas3. rfp (PCR Product) and BBa_R0073 | | show a result for K10_cascade and K10_cas3. rfp (PCR Product) and BBa_R0073 |
| − | (Mnt Promotor) were digested and ligated. <b>19 Eva Patrick</b> Digestion and Ligation | + | (Mnt Promotor) were digested and ligated. <b>19 Eva Patrick</b> Digestion and |
| | + | Ligation |
| | of cascade, cas3 and K10 were repeated with increased amounts of DNA. DH5-Alpha | | of cascade, cas3 and K10 were repeated with increased amounts of DNA. DH5-Alpha |
| | cells were transformed with K10_cascade, K10_cas3 and BBa_R0073(Mnt)_rfp | | cells were transformed with K10_cascade, K10_cas3 and BBa_R0073(Mnt)_rfp |
| Line 10,848: |
Line 10,868: |
| | class="figure-img img-fluid rounded" | | class="figure-img img-fluid rounded" |
| | alt="A generic square placeholder"></a> | | alt="A generic square placeholder"></a> |
| − | <figcaption style="color: #F4F0DE; font-size: small;">Fig. 3. 07/18/19 Gelelctrophoresis of rfp PCR, digests | + | <figcaption style="color: #F4F0DE; font-size: small;">Figure 3: 07/18/19 |
| | + | Gelelctrophoresis of rfp PCR, digests |
| | of K10_cascade and K10_cas3. Gel loaded (from left to right): Ladder 1 | | of K10_cascade and K10_cas3. Gel loaded (from left to right): Ladder 1 |
| | kb, rfp 1, rfp 2, rfp 3, K10_cas3 (no heat inact.), K10_cas3, K10_cascade, | | kb, rfp 1, rfp 2, rfp 3, K10_cas3 (no heat inact.), K10_cas3, K10_cascade, |
| Line 10,874: |
Line 10,895: |
| | <div class="card-body" style="background-color: #232323;"> | | <div class="card-body" style="background-color: #232323;"> |
| | <p style="color: #F4F0DE"> | | <p style="color: #F4F0DE"> |
| − | <b>23 Eva Jakob Patrick</b> Preparation of LB-Medium, LB-Agar and Chloramphenicol plates. | + | <b>23 Eva Jakob Patrick</b> Preparation of LB-Medium, LB-Agar and |
| − | Overnight cultures for K10_cascade, K10_cas3 and BBa_R0073(Mnt)_rfp colonies were | + | Chloramphenicol plates. |
| − | inoculated. After double digestion of BBa_K584000 (AraC Promoter), BBa_K117008 (LsrR Promotor), | + | Overnight cultures for K10_cascade, K10_cas3 and BBa_R0073(Mnt)_rfp colonies |
| − | the fragments ParaC and PLsrR were extracted from the gel. <b>24 Eva Patrick</b> After Miniprep of | + | were |
| − | K10_cascade, K10_cas3 and BBa_R0073(Mnt)_rfp (Qiagen) the plasmids were single digested. Gel | + | inoculated. After double digestion of BBa_K584000 (AraC Promoter), BBa_K117008 |
| − | electrophoresis showed the empty backbone (Fig. 4). The isolated fragments ParaC and PLsrR were | + | (LsrR Promotor), |
| − | ligated into the backbone pSB1K3 after digestion. pSB1K3_ParaC and pSB1K3_PLsrR were transformed | + | the fragments ParaC and PLsrR were extracted from the gel. <b>24 Eva Patrick</b> |
| − | in DH5-Alpha cells. <b>25 Eva</b> Overnight cultures for pSB1K3_ParaC and pSB1K3_PLsrR colonies were | + | After Miniprep of |
| − | inoculated. <b>26 Eva</b> After Miniprep (Qiagen) of pSB1K3_ParaC and pSB1K3_PLsrR, the plasmids were | + | K10_cascade, K10_cas3 and BBa_R0073(Mnt)_rfp (Qiagen) the plasmids were single |
| | + | digested. Gel |
| | + | electrophoresis showed the empty backbone (Fig. 4). The isolated fragments ParaC |
| | + | and PLsrR were |
| | + | ligated into the backbone pSB1K3 after digestion. pSB1K3_ParaC and pSB1K3_PLsrR |
| | + | were transformed |
| | + | in DH5-Alpha cells. <b>25 Eva</b> Overnight cultures for pSB1K3_ParaC and |
| | + | pSB1K3_PLsrR colonies were |
| | + | inoculated. <b>26 Eva</b> After Miniprep (Qiagen) of pSB1K3_ParaC and |
| | + | pSB1K3_PLsrR, the plasmids were |
| | digested. Gel electrophoresis showed successful ligations (Fig. 5). | | digested. Gel electrophoresis showed successful ligations (Fig. 5). |
| | </p> | | </p> |
| Line 10,893: |
Line 10,923: |
| | class="figure-img img-fluid rounded" | | class="figure-img img-fluid rounded" |
| | alt="A generic square placeholder"></a> | | alt="A generic square placeholder"></a> |
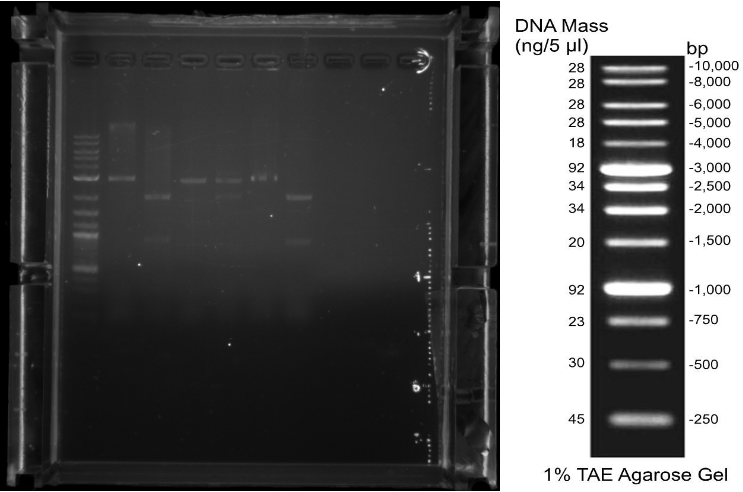
| − | <figcaption style="color: #F4F0DE; font-size: small;">Fig. 4. 07/24/19 Single digestions after Miniprep. | + | <figcaption style="color: #F4F0DE; font-size: small;">Figure 4: 07/24/19 Single |
| | + | digestions after Miniprep. |
| | Gel loaded (from left to right): Ladder 1kb, BBa_R0073(Mnt)_rfp 1.1, | | Gel loaded (from left to right): Ladder 1kb, BBa_R0073(Mnt)_rfp 1.1, |
| | BBa_R0073(Mnt)_rfp 1.2, K10_cas3 1.1-3.2, K10_cascade 1.1-3.2. | | BBa_R0073(Mnt)_rfp 1.2, K10_cas3 1.1-3.2, K10_cascade 1.1-3.2. |
| Line 10,908: |
Line 10,939: |
| | class="figure-img img-fluid rounded" | | class="figure-img img-fluid rounded" |
| | alt="A generic square placeholder image"></a> | | alt="A generic square placeholder image"></a> |
| − | <figcaption style="color: #F4F0DE; font-size: small;">Fig. 5. 07/26/19 Single Digestions after Miniprep. | + | <figcaption style="color: #F4F0DE; font-size: small;">Figure 5: 07/26/19 Single |
| | + | Digestions after Miniprep. |
| | Gel loaded (from left to right): pSB1K3_ParaC 1-8, pSB1K3_PLsrR 1-8, Ladder | | Gel loaded (from left to right): pSB1K3_ParaC 1-8, pSB1K3_PLsrR 1-8, Ladder |
| | 1kb, pSB1K3 2.2-2.3, 3.1-3.2. Bands of ~3 kb suggest successful ligations. | | 1kb, pSB1K3 2.2-2.3, 3.1-3.2. Bands of ~3 kb suggest successful ligations. |
| Line 10,932: |
Line 10,964: |
| | <div class="card-body" style="background-color: #232323;"> | | <div class="card-body" style="background-color: #232323;"> |
| | <p style="color: #F4F0DE"> | | <p style="color: #F4F0DE"> |
| − | <b>30 Patrick, Katharina</b> The team moved into a new laboratory. The synthesized | + | <b>30 Patrick, Katharina</b> The team moved into a new laboratory. The |
| | + | synthesized |
| | sequences (K0, K3, K4, K6, K7, K15) arrived and were aliquoted. Chloramphenicol | | sequences (K0, K3, K4, K6, K7, K15) arrived and were aliquoted. Chloramphenicol |
| − | plates were prepared. cascade, cas3, BBa_I13453 (K10), rfp and BBa_R0073 | + | plates were prepared. cascade, cas3, BBa_I13453 (K10), rfp and BBa_R0073 |
| | (Mnt Promotor) digestions and ligations were repeated and transformed. | | (Mnt Promotor) digestions and ligations were repeated and transformed. |
| − | <b>31 Patrick Jakob</b> Overnight cultures for K10_cas3 colonies were inoculated, | + | <b>31 Patrick Jakob</b> Overnight cultures for K10_cas3 colonies were |
| | + | inoculated, |
| | there was no growth of the other transformations. A PCR (Pfu polymerase) was | | there was no growth of the other transformations. A PCR (Pfu polymerase) was |
| | run on the synthesized constructs, it wasn’t successful. The Minipreps of | | run on the synthesized constructs, it wasn’t successful. The Minipreps of |
| − | pSB1K3_ParaC and pSB1K3_PLsrR were used for retransformation. <b>01 Patrick</b> The | + | pSB1K3_ParaC and pSB1K3_PLsrR were used for retransformation. <b>01 Patrick</b> |
| | + | The |
| | K10_cas3 overnight culture was miniprepped. Overnight cultures for pSB1K3_ParaC, | | K10_cas3 overnight culture was miniprepped. Overnight cultures for pSB1K3_ParaC, |
| − | pSB1K3_PLsrR, as well as K10_cascade and BBa_R0073(Mnt)_rfp (some late colonies grew) | + | pSB1K3_PLsrR, as well as K10_cascade and BBa_R0073(Mnt)_rfp (some late colonies |
| − | were inoculated. <b>02 Patrick</b> Glycerol stocks for pSB1K3_ParaC were prepared, | + | grew) |
| | + | were inoculated. <b>02 Patrick</b> Glycerol stocks for pSB1K3_ParaC were |
| | + | prepared, |
| | there was no growth in pSB1K3_PLsrR overnight cultures. After Miniprep of | | there was no growth in pSB1K3_PLsrR overnight cultures. After Miniprep of |
| | rpSB1K3_ParaC, K10_cascade and BBa_R0073(Mnt)_rfp the plasmids (also K10_cas3) | | rpSB1K3_ParaC, K10_cascade and BBa_R0073(Mnt)_rfp the plasmids (also K10_cas3) |
| Line 10,957: |
Line 10,994: |
| | class="figure-img img-fluid rounded" | | class="figure-img img-fluid rounded" |
| | alt="A generic square placeholder"></a> | | alt="A generic square placeholder"></a> |
| − | <figcaption style="color: #F4F0DE; font-size: small;">Fig. 6. 08/02/19 Digestions after Miniprep. Gel loaded | + | <figcaption style="color: #F4F0DE; font-size: small;">Figure 6: 08/02/19 |
| | + | Digestions after Miniprep. Gel loaded |
| | (from left to right): Ladder 1 kb, K10_cas3 1-10, K10_cascade 1-4, | | (from left to right): Ladder 1 kb, K10_cas3 1-10, K10_cascade 1-4, |
| | BBa_R0073(Mnt)_rfp 1-2, positive control, pSB1K3_ParaC. Negative results | | BBa_R0073(Mnt)_rfp 1-2, positive control, pSB1K3_ParaC. Negative results |
| Line 10,983: |
Line 11,021: |
| | <div class="card-body" style="background-color: #232323;"> | | <div class="card-body" style="background-color: #232323;"> |
| | <p style="color: #F4F0DE"> | | <p style="color: #F4F0DE"> |
| − | <b>05 Patrick Eva Antonia</b> PCR (Q5 polymerase) and PCR (Paq polymerase) was run on the | + | <b>05 Patrick Eva Antonia</b> PCR (Q5 polymerase) and PCR (Paq polymerase) was |
| − | synthesized constructs, they weren’t successful. cascade and cas3 were successfully | + | run on the |
| − | amplified via PCR(Q5). <b>06 Eva Patrick Antonia</b> A gradient PCR (Q5) was run on the constructs. | + | synthesized constructs, they weren’t successful. cascade and cas3 were |
| − | It was discovered that the currently used agarose was falsely prepared with water instead | + | successfully |
| − | of TAE-Buffer and was discarded. cascade, cas3 and BBa_I13453 (K10) were digested and ligated. | + | amplified via PCR(Q5). <b>06 Eva Patrick Antonia</b> A gradient PCR (Q5) was run |
| − | <b>07 Patrick Eva Luzi Antonia</b> A new PCR (Q5) was run on the synthesized constructs, | + | on the constructs. |
| | + | It was discovered that the currently used agarose was falsely prepared with |
| | + | water instead |
| | + | of TAE-Buffer and was discarded. cascade, cas3 and BBa_I13453 (K10) were |
| | + | digested and ligated. |
| | + | <b>07 Patrick Eva Luzi Antonia</b> A new PCR (Q5) was run on the synthesized |
| | + | constructs, |
| | gel electrophoresis suggested positive amplification (Fig. 7). By amplification | | gel electrophoresis suggested positive amplification (Fig. 7). By amplification |
| | with accordingly designed primer, K0 has been added a TetR promoter and is now | | with accordingly designed primer, K0 has been added a TetR promoter and is now |
| − | the construct K1. Due to a technical malfunction in the building further work was delayed. | + | the construct K1. Due to a technical malfunction in the building further work |
| | + | was delayed. |
| | </p> | | </p> |
| | <figure> | | <figure> |
| Line 11,001: |
Line 11,046: |
| | class="figure-img img-fluid rounded" | | class="figure-img img-fluid rounded" |
| | alt="A generic square placeholder"></a> | | alt="A generic square placeholder"></a> |
| − | <figcaption style="color: #F4F0DE; font-size: small;">Fig. 7. 08/08/19 PCR of synthesized constructs. | + | <figcaption style="color: #F4F0DE; font-size: small;">Figure 7: 08/08/19 PCR of |
| | + | synthesized constructs. |
| | Gel loaded (from left to right): Ladder 1kb, K1 (expected at 660bp), | | Gel loaded (from left to right): Ladder 1kb, K1 (expected at 660bp), |
| | K3 (expected at 1 kb), K4 (expected at 1 kb), K6 (expected at 1.5 kb), | | K3 (expected at 1 kb), K4 (expected at 1 kb), K6 (expected at 1.5 kb), |
| Line 11,026: |
Line 11,072: |
| | <div class="card-body" style="background-color: #232323;"> | | <div class="card-body" style="background-color: #232323;"> |
| | <p style="color: #F4F0DE"> | | <p style="color: #F4F0DE"> |
| − | <b>12 Patrick Katharina</b> After respective digestion and DNA clean up, pSBC13_K1, | + | <b>12 Patrick Katharina</b> After respective digestion and DNA clean up, |
| | + | pSBC13_K1, |
| | pSBC13_K3, pSBC13_K6, K5_K4, K10_cas3, K10_cascade and BBa_R0073(Mnt)_rfp were | | pSBC13_K3, pSBC13_K6, K5_K4, K10_cas3, K10_cascade and BBa_R0073(Mnt)_rfp were |
| − | ligated. <b>13 Patrick Eva Antonia Marie</b> DH5-Alpha cells were transformed with | + | ligated. <b>13 Patrick Eva Antonia Marie</b> DH5-Alpha cells were transformed |
| − | Yesterday’s ligations. <b>14 Luzie Marie Eva</b> A colony PCR (Taq polymerase) was | + | with |
| | + | Yesterday’s ligations. <b>14 Luzie Marie Eva</b> A colony PCR (Taq polymerase) |
| | + | was |
| | run on pSBC13_K1, but the gel electrophoresis was negative (Fig. 8). Overnight | | run on pSBC13_K1, but the gel electrophoresis was negative (Fig. 8). Overnight |
| | cultures of pSBC13_K1, pSBC13_K3, pSBC13_K6, K5_K4, K10_cas3 and K10_cascade | | cultures of pSBC13_K1, pSBC13_K3, pSBC13_K6, K5_K4, K10_cas3 and K10_cascade |
| − | colonies were inoculated. <b>15 Marie Patrick Eva</b> After Miniprep the plasmids were | + | colonies were inoculated. <b>15 Marie Patrick Eva</b> After Miniprep the |
| − | double digested. Gel electrophoresis suggested successful ligations of pSBC13_K6, | + | plasmids were |
| − | K10_cas3, K10_cascade and BBa_R0073(Mnt)_rfp (Fig. 9). <b>16 Patrick</b> Miniprep of | + | double digested. Gel electrophoresis suggested successful ligations of |
| | + | pSBC13_K6, |
| | + | K10_cas3, K10_cascade and BBa_R0073(Mnt)_rfp (Fig. 9). <b>16 Patrick</b> |
| | + | Miniprep of |
| | pSBC13_K1 was digested but gel electrophoresis was negative. cascade and cas3 | | pSBC13_K1 was digested but gel electrophoresis was negative. cascade and cas3 |
| | were amplified by PCR (Q5 polymerase). The synthesized construct K2 arrived, | | were amplified by PCR (Q5 polymerase). The synthesized construct K2 arrived, |
| Line 11,047: |
Line 11,099: |
| | class="figure-img img-fluid rounded" | | class="figure-img img-fluid rounded" |
| | alt="A generic square placeholder"></a> | | alt="A generic square placeholder"></a> |
| − | <figcaption style="color: #F4F0DE; font-size: small;">Fig. 8. 08/14/19 Colony PCR of pSB1C3_K1 transformation. | + | <figcaption style="color: #F4F0DE; font-size: small;">Figure 8: 08/14/19 Colony |
| | + | PCR of pSB1C3_K1 transformation. |
| | Gel loaded (from left to right): Ladder 1kb, pSB1C3_K0 1-10. | | Gel loaded (from left to right): Ladder 1kb, pSB1C3_K0 1-10. |
| | </figcaption> | | </figcaption> |
| Line 11,059: |
Line 11,112: |
| | class="figure-img img-fluid rounded" | | class="figure-img img-fluid rounded" |
| | alt="A generic square placeholder image"></a> | | alt="A generic square placeholder image"></a> |
| − | <figcaption style="color: #F4F0DE; font-size: small;">Fig. 9. 08/15/19 Digestions after Miniprep. Gel loaded | + | <figcaption style="color: #F4F0DE; font-size: small;">Figure 9: 08/15/19 |
| − | (from left to right): Ladder 1kb, pSB1C3_K6 1, empty, pSB1C3_K6 2, pSB1C3_K3 1-2, | + | Digestions after Miniprep. Gel loaded |
| | + | (from left to right): Ladder 1kb, pSB1C3_K6 1, empty, pSB1C3_K6 2, pSB1C3_K3 |
| | + | 1-2, |
| | K10_cas3 1-2, K10_cascade 1-2, BBa_R0073(Mnt)_rfp 1-4. | | K10_cas3 1-2, K10_cascade 1-2, BBa_R0073(Mnt)_rfp 1-4. |
| − | K6 expected at ~2 kb and 1.5 kb, K3 expected at ~2 kb and 1 kb, cas3 expected at ~2.4 kb, ~2 kb and ~0.3 kb, cascade | + | K6 expected at ~2 kb and 1.5 kb, K3 expected at ~2 kb and 1 kb, cas3 |
| | + | expected at ~2.4 kb, ~2 kb and ~0.3 kb, cascade |
| | expected at ~5.1 kb, ~1,2 kb and ~0.3 kb, rfp expected at ~2 kb and ~1 kb. | | expected at ~5.1 kb, ~1,2 kb and ~0.3 kb, rfp expected at ~2 kb and ~1 kb. |
| | </figcaption> | | </figcaption> |
| Line 11,086: |
Line 11,142: |
| | <div class="card-body" style="background-color: #232323;"> | | <div class="card-body" style="background-color: #232323;"> |
| | <p style="color: #F4F0DE"> | | <p style="color: #F4F0DE"> |
| − | <b>19 Patrick Jakob Eva</b> After respective digestion and DNA clean up, pSB1C3_K1, | + | <b>19 Patrick Jakob Eva</b> After respective digestion and DNA clean up, |
| | + | pSB1C3_K1, |
| | pSB1C3_K3, pSB1C3_K7 and K5_K4 were ligated. The BBa_R0073(Mnt)_rfp plasmid was | | pSB1C3_K3, pSB1C3_K7 and K5_K4 were ligated. The BBa_R0073(Mnt)_rfp plasmid was |
| − | digested to isolate PMnt_rfp by gel extraction (Fig. 10). The backbone pSB1K3 was | + | digested to isolate PMnt_rfp by gel extraction (Fig. 10). The backbone pSB1K3 |
| − | digested with respective enzymes and pSB1K3_PMnt_rfp was ligated. K1, K2, K3, K4, | + | was |
| − | K7 and K15 were amplified via PCR (Q5 polymerase) with a new programm. <b>20 Eva Patrick</b> | + | digested with respective enzymes and pSB1K3_PMnt_rfp was ligated. K1, K2, K3, |
| − | Last week’s pSBC13_K6, K10_cas3 and K10_cascade were sent off to Sanger sequencing. | + | K4, |
| − | DH5-Alpha cells were transformed with yesterday’s ligations as well as retransformed | + | K7 and K15 were amplified via PCR (Q5 polymerase) with a new programm. <b>20 Eva |
| − | with K10_cascade . <b>21 Marie Zoe Patrick</b> A new batch of competent cells was prepared | + | Patrick</b> |
| − | and overnight cultures of pSB1C3_K1, pSB1C3_K7, K5_K4 and K10_cascade inoculated. | + | Last week’s pSBC13_K6, K10_cas3 and K10_cascade were sent off to Sanger |
| − | <b>22 Patrick Eva</b> Sanger Sequencing was positive on K10_cascade. Chloramphenicol and | + | sequencing. |
| − | Kanamycin plates were prepared. After Miniprep of pSB1C3_K1, pSB1C3_K7, K5_K4 and | + | DH5-Alpha cells were transformed with yesterday’s ligations as well as |
| | + | retransformed |
| | + | with K10_cascade . <b>21 Marie Zoe Patrick</b> A new batch of competent cells |
| | + | was prepared |
| | + | and overnight cultures of pSB1C3_K1, pSB1C3_K7, K5_K4 and K10_cascade |
| | + | inoculated. |
| | + | <b>22 Patrick Eva</b> Sanger Sequencing was positive on K10_cascade. |
| | + | Chloramphenicol and |
| | + | Kanamycin plates were prepared. After Miniprep of pSB1C3_K1, pSB1C3_K7, K5_K4 |
| | + | and |
| | K10_cascade the plasmids were digested and run on a gel. Digestions of pSB1C3_K1 | | K10_cascade the plasmids were digested and run on a gel. Digestions of pSB1C3_K1 |
| − | and K5_K4 were negative. The new competent cells were transformed with pSB1C3_K3, | + | and K5_K4 were negative. The new competent cells were transformed with |
| − | pSB1K3_PMnt_rfp and iGEM competent cell test plasmids. <b>23 Marie Patrick</b> The | + | pSB1C3_K3, |
| | + | pSB1K3_PMnt_rfp and iGEM competent cell test plasmids. <b>23 Marie Patrick</b> |
| | + | The |
| | constructs K1, K2, K3, K4, K7 and K15 were amplified by PCR (Q5 polymerase). | | constructs K1, K2, K3, K4, K7 and K15 were amplified by PCR (Q5 polymerase). |
| − | Double digestion of pSB1C3_K7 and K10_cascade was repeated. The gel electrophoresis | + | Double digestion of pSB1C3_K7 and K10_cascade was repeated. The gel |
| − | suggested successful ligation of K7, the result for K10_cascade was unclear (Fig. 11). | + | electrophoresis |
| | + | suggested successful ligation of K7, the result for K10_cascade was unclear |
| | + | (Fig. 11). |
| | </p> | | </p> |
| | <figure> | | <figure> |
| Line 11,113: |
Line 11,183: |
| | class="figure-img img-fluid rounded" | | class="figure-img img-fluid rounded" |
| | alt="A generic square placeholder"></a> | | alt="A generic square placeholder"></a> |
| − | <figcaption style="color: #F4F0DE; font-size: small;">Fig. 10. 08/19/19 Digestion of BBa_R0073(Mnt)_rfp for | + | <figcaption style="color: #F4F0DE; font-size: small;">Figure 10: 08/19/19 |
| − | gel extraction. Gel loaded (from left to right): Ladder 1 kb, BBa_R0073(Mnt)_rfp. | + | Digestion of BBa_R0073(Mnt)_rfp for |
| | + | gel extraction. Gel loaded (from left to right): Ladder 1 kb, |
| | + | BBa_R0073(Mnt)_rfp. |
| | Desired fragment at ~0.5 kb. | | Desired fragment at ~0.5 kb. |
| | </figcaption> | | </figcaption> |
| Line 11,126: |
Line 11,198: |
| | class="figure-img img-fluid rounded" | | class="figure-img img-fluid rounded" |
| | alt="A generic square placeholder image"></a> | | alt="A generic square placeholder image"></a> |
| − | <figcaption style="color: #F4F0DE; font-size: small;">Fig. 11. Digestion after Miniprep. Gel loaded (from left to right): | + | <figcaption style="color: #F4F0DE; font-size: small;">Figure 11: Digestion after |
| | + | Miniprep. Gel loaded (from left to right): |
| | Ladder 1 kb, pSB1C3_K7 1-2, K10_cascade 1-5, K10_cas3. | | Ladder 1 kb, pSB1C3_K7 1-2, K10_cascade 1-5, K10_cas3. |
| | </figcaption> | | </figcaption> |
| Line 11,151: |
Line 11,224: |
| | <div class="card-body" style="background-color: #232323;"> | | <div class="card-body" style="background-color: #232323;"> |
| | <p style="color: #F4F0DE"> | | <p style="color: #F4F0DE"> |
| − | <b>26 Patrick</b> A new PCR (Q5 polymerase) was run on the constructs and the successfully | + | <b>26 Patrick</b> A new PCR (Q5 polymerase) was run on the constructs and the |
| − | amplified DNA purified. K10_cascade and pSB1C3_K6 were retransformed. Inoculation of | + | successfully |
| − | overnight cultures for K10_cascade and K10_cas3 old colonies. <b>27 Patrick Eva Jakob Antonia</b> | + | amplified DNA purified. K10_cascade and pSB1C3_K6 were retransformed. |
| − | Miniprep of K10_cascade and K10_cas3 followed by digestion and gel electrophoresis, | + | Inoculation of |
| | + | overnight cultures for K10_cascade and K10_cas3 old colonies. <b>27 Patrick Eva |
| | + | Jakob Antonia</b> |
| | + | Miniprep of K10_cascade and K10_cas3 followed by digestion and gel |
| | + | electrophoresis, |
| | which suggested successful ligation of K10_cascade, K10_cas3 is XY (Fig. 12). | | which suggested successful ligation of K10_cascade, K10_cas3 is XY (Fig. 12). |
| − | All synthesized constructs (K0-K15) were digested and ligated with their respective | + | All synthesized constructs (K0-K15) were digested and ligated with their |
| | + | respective |
| | backbone. Overnight cultures of K10_cascade and pSB1C3_K6 retransformations were | | backbone. Overnight cultures of K10_cascade and pSB1C3_K6 retransformations were |
| − | inoculated. <b>28 Patrick Eva Antonia</b> The BBa_R0073(Mnt)_rfp plasmid was digested to | + | inoculated. <b>28 Patrick Eva Antonia</b> The BBa_R0073(Mnt)_rfp plasmid was |
| | + | digested to |
| | isolate PMnt_rfp and the backbone pSB1K3 was digested with respective enzymes. | | isolate PMnt_rfp and the backbone pSB1K3 was digested with respective enzymes. |
| | pSB1K3_PMnt_rfp was ligated. DH5-Alpha cells were transformed with yesterday’s | | pSB1K3_PMnt_rfp was ligated. DH5-Alpha cells were transformed with yesterday’s |
| − | construct (K0-K15) ligations. Overnight cultures of K10_cascade and pSB1C3_K6 were | + | construct (K0-K15) ligations. Overnight cultures of K10_cascade and pSB1C3_K6 |
| | + | were |
| | miniprepped and digested. The gel electrophoresis showed bands suggesting a | | miniprepped and digested. The gel electrophoresis showed bands suggesting a |
| | successful ligation of K10_cascade and pSB1C3_K6 (Fig. 13). The synthesized | | successful ligation of K10_cascade and pSB1C3_K6 (Fig. 13). The synthesized |
| − | constructs were amplified via PCR (Q5 polymerase) for our Experimenta collaboration. | + | constructs were amplified via PCR (Q5 polymerase) for our Experimenta |
| | + | collaboration. |
| | <b>29 Eva Antonia Patrick</b> A colony PCR (Q5) was run on K5_K4, the other | | <b>29 Eva Antonia Patrick</b> A colony PCR (Q5) was run on K5_K4, the other |
| | transformations didn’t show growth. Gel electrophoresis of the PCR didn’t | | transformations didn’t show growth. Gel electrophoresis of the PCR didn’t |
| − | show a positive result. To amplify pSB1C3, a biobrick of the iGEM distribution | + | show a positive result. To amplify pSB1C3, a biobrick of the iGEM distribution |
| | plates was diluted and a PCR (Q5 polymerase) was run. The constructs (K0-K15) | | plates was diluted and a PCR (Q5 polymerase) was run. The constructs (K0-K15) |
| | were digested. K0, K2, K3, K15 were ligated with their backbone pSB1C3. K4 was | | were digested. K0, K2, K3, K15 were ligated with their backbone pSB1C3. K4 was |
| | ligated with BBa_K091001 (K5) and K7 was ligated with pSB1C3_K6. The work on the | | ligated with BBa_K091001 (K5) and K7 was ligated with pSB1C3_K6. The work on the |
| | cell penetrating peptides (CPPs) was started by amplifying gfp and synb1 via PCR | | cell penetrating peptides (CPPs) was started by amplifying gfp and synb1 via PCR |
| − | (Q5 polymerase) (Fig. CPP 1). Primer were dissolved to 100 µM. <b>30 Antonia Eva Katharina</b> | + | (Q5 polymerase) (Fig. CPP 1). Primer were dissolved to 100 µM. <b>30 Antonia Eva |
| − | The colony PCR for K4_K5 was repeated, but remained negative. PCR (Q5 polymerase) | + | Katharina</b> |
| | + | The colony PCR for K4_K5 was repeated, but remained negative. PCR (Q5 |
| | + | polymerase) |
| | was run on CPPs. Gel electrophoreses of the PCR showed double bands. | | was run on CPPs. Gel electrophoreses of the PCR showed double bands. |
| | </p> | | </p> |
| Line 11,184: |
Line 11,267: |
| | class="figure-img img-fluid rounded" | | class="figure-img img-fluid rounded" |
| | alt="Figure 12"></a> | | alt="Figure 12"></a> |
| − | <figcaption style="color: #F4F0DE; font-size: small;">Fig. 12. 08/27/19 Digestions after Miniprep. Gel loaded | + | <figcaption style="color: #F4F0DE; font-size: small;">Figure 12: 08/27/19 |
| − | (from left to right): ladder 1 kb, K10_cascade 1-7, K10_cas3 1-7. K10_cascade | + | Digestions after Miniprep. Gel loaded |
| | + | (from left to right): ladder 1 kb, K10_cascade 1-7, K10_cas3 1-7. |
| | + | K10_cascade |
| | expected at ~6.6 kb, K10_cas3 expected at ~4.9 kb. | | expected at ~6.6 kb, K10_cas3 expected at ~4.9 kb. |
| | </figcaption> | | </figcaption> |
| Line 11,197: |
Line 11,282: |
| | class="figure-img img-fluid rounded" | | class="figure-img img-fluid rounded" |
| | alt="Figure 13"></a> | | alt="Figure 13"></a> |
| − | <figcaption style="color: #F4F0DE; font-size: small;">Fig. 13. 08/28/19 Digestions after Miniprep. Gel loaded | + | <figcaption style="color: #F4F0DE; font-size: small;">Figure 13: 08/28/19 |
| − | (from left to right): Ladder 1 kb, K10_cascade 1, empty, K10_cascade 2-4, pSB1C3_K6 | + | Digestions after Miniprep. Gel loaded |
| − | 1-4. K10_cascade expected at ~5.1 kb, ~1,2 kb and ~0.3 kb, pSB1C3_K6 expected at ~ 2 kb | + | (from left to right): Ladder 1 kb, K10_cascade 1, empty, K10_cascade 2-4, |
| | + | pSB1C3_K6 |
| | + | 1-4. K10_cascade expected at ~5.1 kb, ~1,2 kb and ~0.3 kb, pSB1C3_K6 |
| | + | expected at ~ 2 kb |
| | and ~1.6 kb. | | and ~1.6 kb. |
| | </figcaption> | | </figcaption> |
| Line 11,209: |
Line 11,297: |
| | | | |
| | | | |
| − | <div id="accordion9">
| + | <div id="accordion9"> |
| | <div class="card"> | | <div class="card"> |
| | <div class="card-header" id="headingNine"> | | <div class="card-header" id="headingNine"> |
| Line 11,225: |
Line 11,313: |
| | <div class="card-body" style="background-color: #232323;"> | | <div class="card-body" style="background-color: #232323;"> |
| | <p style="color: #F4F0DE"> | | <p style="color: #F4F0DE"> |
| − | <b>02 Lina Antonia Luzi Jakob Eva Famke</b> As preliminary testing, a growth curve of | + | <b>02 Lina Antonia Luzi Jakob Eva Famke</b> As preliminary testing, a growth |
| − | <i>E. coli</i> Nissle at 37°C, pH 7, was generated. DH5-Alpha cells were transformed | + | curve of |
| | + | <i>E. coli</i> Nissle at 37°C, pH 7, was generated. DH5-Alpha cells were |
| | + | transformed |
| | with the ligations of constructs (K0-K15) as well as pSB1K3_PMnt_rfp and the | | with the ligations of constructs (K0-K15) as well as pSB1K3_PMnt_rfp and the |
| − | distribution kit biobrick. The CPPs tat-lk15, tat, yta2, tp10, pVec and penetratin | + | distribution kit biobrick. The CPPs tat-lk15, tat, yta2, tp10, pVec and |
| − | were amplified by PCR (HotStart Polymerase). The gel electrophoresis showed double | + | penetratin |
| − | bands (Fig. CPP 2). <b>03/04 Lina</b> Growth curves of <i>E. coli</i> Nissle (EcN) for different | + | were amplified by PCR (HotStart Polymerase). The gel electrophoresis showed |
| − | temperatures were created. EcN cultures were inoculated with an overnight culture | + | double |
| − | and grown for 120 minutes at 37°C. Next, temperature was changed to 0°C, 4°C, 8°C | + | bands (Fig. CPP 2). <b>03/04 Lina</b> Growth curves of <i>E. coli</i> Nissle |
| − | and 25°C, while one control was kept at 37°C. 37°C and 8°C were chosen for sample | + | (EcN) for different |
| | + | temperatures were created. EcN cultures were inoculated with an overnight |
| | + | culture |
| | + | and grown for 120 minutes at 37°C. Next, temperature was changed to 0°C, 4°C, |
| | + | 8°C |
| | + | and 25°C, while one control was kept at 37°C. 37°C and 8°C were chosen for |
| | + | sample |
| | preparation for RNA-Seq. EcN was grown 120 min, after being inoculated from one | | preparation for RNA-Seq. EcN was grown 120 min, after being inoculated from one |
| − | overnight culture, at pH7. Then, media was changed to the respective pH (6, 5, 4, 3, 2, 1) | + | overnight culture, at pH7. Then, media was changed to the respective pH (6, 5, |
| − | and growth was observed. Growth in pH4 was replicated. EcN was grown in different | + | 4, 3, 2, 1) |
| | + | and growth was observed. Growth in pH4 was replicated. EcN was grown in |
| | + | different |
| | concentrations of Hydrogen Peroxide (1 µM, 10 µM, 25 µM, 100 µM), infused after | | concentrations of Hydrogen Peroxide (1 µM, 10 µM, 25 µM, 100 µM), infused after |
| − | two hours of growth at 37°C, pH7 in LB medium. EcN, inoculated from the same overnight | + | two hours of growth at 37°C, pH7 in LB medium. EcN, inoculated from the same |
| − | culture, were grown under four anaerobic conditions (LB, mGAM anaerobic, mGAM+1.3 | + | overnight |
| − | mM Metformin, mGAM+0,25mM cholic acid) at 37°C, pH7. EcN aerobic growth in LB was | + | culture, were grown under four anaerobic conditions (LB, mGAM anaerobic, |
| | + | mGAM+1.3 |
| | + | mM Metformin, mGAM+0,25mM cholic acid) at 37°C, pH7. EcN aerobic growth in LB |
| | + | was |
| | used as control. Samples for RNA sequencing were taken after 270 minutes for all | | used as control. Samples for RNA sequencing were taken after 270 minutes for all |
| − | anaerobic conditions, since the increase of pressure within the flasks indicated a | + | anaerobic conditions, since the increase of pressure within the flasks indicated |
| | + | a |
| | change of metabolism of EcN to fermentation processes. Aerobic LB growth samples | | change of metabolism of EcN to fermentation processes. Aerobic LB growth samples |
| | were taken at OD600=1.0. | | were taken at OD600=1.0. |
| Line 11,249: |
Line 11,351: |
| | EcN, inoculated from the same overnight culture, were grown in mGAM medium under | | EcN, inoculated from the same overnight culture, were grown in mGAM medium under |
| | anaerobic conditions at 37°C, pH 7 supplemented with either 50% supernatant of | | anaerobic conditions at 37°C, pH 7 supplemented with either 50% supernatant of |
| − | different species or 50% of water as a control for depleted nutrients. Supernatant of Bacteroides | + | different species or 50% of water as a control for depleted nutrients. |
| − | thetaiotaomicron, Prevotella copri, Ruminococcus gnavus, Bifidobacterium adolescentis and | + | Supernatant of Bacteroides |
| − | Clostridium difficile was previously produced by Dr. Lisa Maier. Bacteroides samples were | + | thetaiotaomicron, Prevotella copri, Ruminococcus gnavus, Bifidobacterium |
| − | taken for RNA sequencing after 225 minutes. EcN were lastly grown in the growth medium provided by | + | adolescentis and |
| | + | Clostridium difficile was previously produced by Dr. Lisa Maier. Bacteroides |
| | + | samples were |
| | + | taken for RNA sequencing after 225 minutes. EcN were lastly grown in the growth |
| | + | medium provided by |
| | iGEM Stuttgart, in comparison to LB medium. Overnight cultures were used for | | iGEM Stuttgart, in comparison to LB medium. Overnight cultures were used for |
| | inoculation of three samples with 5ml each and OD600 was measured every 45 mins. | | inoculation of three samples with 5ml each and OD600 was measured every 45 mins. |
| − | For the dryfreezed samples, 9 µL skim milk were added to 1 µg wet weight of bacteria, | + | For the dryfreezed samples, 9 µL skim milk were added to 1 µg wet weight of |
| − | frozen at -80°C for 2 h, 42 h at -20°C and 2h at 25°C. Afterwards they were resuspended in LB. | + | bacteria, |
| | + | frozen at -80°C for 2 h, 42 h at -20°C and 2h at 25°C. Afterwards they were |
| | + | resuspended in LB. |
| | </p> | | </p> |
| | <p> | | <p> |
| − | All OD600 measurements were conducted with the Nanospectrometre by Implen. For the | + | All OD600 measurements were conducted with the Nanospectrometre by Implen. For |
| − | results check <a href="https://2019.igem.org/Team:Tuebingen/Nissle" onclick='window.open("https://2019.igem.org/Team:Tuebingen/Nissle","_self");'>Project/ Nissle</a>. | + | the |
| − | <b>04 Eva Antonia</b> A batch of competent cells was prepared. pSB1C3_K0, pSB1C3_K2, | + | results check <a href="https://2019.igem.org/Team:Tuebingen/Nissle" |
| − | pSB1C3_K3, pSB1C3_K15, pSB1C3_K6_K7, K5_K4 and pSB1K3_PMnt_rfp colonies were analyzed | + | onclick='window.open("https://2019.igem.org/Team:Tuebingen/Nissle","_self");'>Project/ |
| − | by colony PCR(Q5). Gel electrophoresis didn’t show positive result. <b>05 Eva Antonia</b> | + | Nissle</a>. |
| | + | <b>04 Eva Antonia</b> A batch of competent cells was prepared. pSB1C3_K0, |
| | + | pSB1C3_K2, |
| | + | pSB1C3_K3, pSB1C3_K15, pSB1C3_K6_K7, K5_K4 and pSB1K3_PMnt_rfp colonies were |
| | + | analyzed |
| | + | by colony PCR(Q5). Gel electrophoresis didn’t show positive result. <b>05 Eva |
| | + | Antonia</b> |
| | Further gel electrophoresis of yesterday’s colony PCR remained negative. | | Further gel electrophoresis of yesterday’s colony PCR remained negative. |
| | Digestions and ligations of constructs were repeated. Overnight cultures of | | Digestions and ligations of constructs were repeated. Overnight cultures of |
| − | distribution kit biobrick colonies were inoculated. <b>06 Marie</b> Miniprep of distribution kit biobrick. | + | distribution kit biobrick colonies were inoculated. <b>06 Marie</b> Miniprep of |
| | + | distribution kit biobrick. |
| | </p> | | </p> |
| | <figure> | | <figure> |
| Line 11,276: |
Line 11,391: |
| | class="figure-img img-fluid rounded" | | class="figure-img img-fluid rounded" |
| | alt="Figure 14"></a> | | alt="Figure 14"></a> |
| − | <figcaption style="color: #F4F0DE; font-size: small;">Fig. CPP 2. 09/02/19 PCR of CPP parts. Gel loaded | + | <figcaption style="color: #F4F0DE; font-size: small;">Figure CPP 2: 09/02/19 PCR |
| | + | of CPP parts. Gel loaded |
| | (from left to right): Ladder 1 kb, pVec (expected at ~100 bp), tat | | (from left to right): Ladder 1 kb, pVec (expected at ~100 bp), tat |
| − | (expected at ~70 bp), tat-lk15 (expected at ~120 bp), tp10 (expected at ~90 bp), | + | (expected at ~70 bp), tat-lk15 (expected at ~120 bp), tp10 (expected at ~90 |
| | + | bp), |
| | yta2 (expected at ~90 bp). | | yta2 (expected at ~90 bp). |
| | </figcaption> | | </figcaption> |
| Line 11,287: |
Line 11,404: |
| | </div> | | </div> |
| | | | |
| − | <div id="accordion10">
| + | <div id="accordion10"> |
| | <div class="card"> | | <div class="card"> |
| | <div class="card-header" id="headingTen"> | | <div class="card-header" id="headingTen"> |
| Line 11,305: |
Line 11,422: |
| | (Q5 polymerase) using primers designed for Gibson assembly, the PCR didn’t work. | | (Q5 polymerase) using primers designed for Gibson assembly, the PCR didn’t work. |
| | After digestion of the CPPs and their backbones, the plasmids pET-28b(+)_synb1 | | After digestion of the CPPs and their backbones, the plasmids pET-28b(+)_synb1 |
| − | as well as pMiniT2.0_pVec and pMiniT2.0_tat-lk15 were ligated. <b>11 Eva Antonia</b> | + | as well as pMiniT2.0_pVec and pMiniT2.0_tat-lk15 were ligated. <b>11 Eva |
| − | A gradient PCR of the constructs was run with Gibson primers, gel electrophoresis | + | Antonia</b> |
| − | suggested successful amplification of K1, K2 and K15 (Fig. 14). <b>12 Eva Antonia</b> | + | A gradient PCR of the constructs was run with Gibson primers, gel |
| | + | electrophoresis |
| | + | suggested successful amplification of K1, K2 and K15 (Fig. 14). <b>12 Eva |
| | + | Antonia</b> |
| | Gibson-PCR of K3, K4, K6, and K7 was successfully repeated. For Gibson assembly | | Gibson-PCR of K3, K4, K6, and K7 was successfully repeated. For Gibson assembly |
| | the backbones pSB1C3 and BBa_K091001 (K5) were single digested. pSB1C3_K1_K15, | | the backbones pSB1C3 and BBa_K091001 (K5) were single digested. pSB1C3_K1_K15, |
| − | pSB1C3_K2_K3, K5_K4 and pSB1C3_K6_K7 were assembled and transformed. Additionally, | + | pSB1C3_K2_K3, K5_K4 and pSB1C3_K6_K7 were assembled and transformed. |
| − | pET-28b(+)_synb1 was transformed. <b>13 Eva Antonia</b> The transformations didn’t | + | Additionally, |
| | + | pET-28b(+)_synb1 was transformed. <b>13 Eva Antonia</b> The transformations |
| | + | didn’t |
| | show colonies so the Gibson assembly was repeated and transformed with provided | | show colonies so the Gibson assembly was repeated and transformed with provided |
| − | NovaBlue™ competent cells. Colonies for pET-28b(+) were picked and an over-weekend | + | NovaBlue™ competent cells. Colonies for pET-28b(+) were picked and an |
| | + | over-weekend |
| | culture (RT) inoculated. The ligations of pMiniT2.0_pVec and pMiniT2.0_tat-lk15 | | culture (RT) inoculated. The ligations of pMiniT2.0_pVec and pMiniT2.0_tat-lk15 |
| | were transformed in DH5-Alpha cells. | | were transformed in DH5-Alpha cells. |
| Line 11,325: |
Line 11,448: |
| | class="figure-img img-fluid rounded" | | class="figure-img img-fluid rounded" |
| | alt="Figure 14"></a> | | alt="Figure 14"></a> |
| − | <figcaption style="color: #F4F0DE; font-size: small;">Fig. 14. 09/11/19 Gibson PCR. Gel loaded (from left to right): Ladder 1 kb, empty, K15 1-3, K1 1-2, empty, K1 3-5, K2 1-5. K15 expected at ~1 kb, K1 expected at ~0.5 kb, K2 expected at ~1 kb. | + | <figcaption style="color: #F4F0DE; font-size: small;">Figure 14: 09/11/19 Gibson |
| | + | PCR. Gel loaded (from left to right): Ladder 1 kb, empty, K15 1-3, K1 1-2, |
| | + | empty, K1 3-5, K2 1-5. K15 expected at ~1 kb, K1 expected at ~0.5 kb, K2 |
| | + | expected at ~1 kb. |
| | </figcaption> | | </figcaption> |
| | </figure> | | </figure> |
| Line 11,333: |
Line 11,459: |
| | </div> | | </div> |
| | | | |
| − | <div id="accordion11">
| + | <div id="accordion11"> |
| | <div class="card"> | | <div class="card"> |
| | <div class="card-header" id="headingEleven"> | | <div class="card-header" id="headingEleven"> |
| Line 11,351: |
Line 11,477: |
| | growth so they were diluted by streaking. The over-weekend cultures of | | growth so they were diluted by streaking. The over-weekend cultures of |
| | pET-28b(+)_synb1 were miniprepped. For pMiniT2.0_pVec and pMiniT2.0_tat-lk15 | | pET-28b(+)_synb1 were miniprepped. For pMiniT2.0_pVec and pMiniT2.0_tat-lk15 |
| − | colonies were picked and overnight cultures inoculated. <b>17 Marie Eva Antonia</b> | + | colonies were picked and overnight cultures inoculated. <b>17 Marie Eva |
| | + | Antonia</b> |
| | The BBa_R0073(Mnt)_rfp plasmid was digested to isolate PMnt_rfp and the backbone | | The BBa_R0073(Mnt)_rfp plasmid was digested to isolate PMnt_rfp and the backbone |
| | pSB1K3 was digested with respective enzymes. pSB1K3_PMnt_rfp was ligated. | | pSB1K3 was digested with respective enzymes. pSB1K3_PMnt_rfp was ligated. |
| | The dilution streaks of Novablue™ Gibson transformations were diluted again. | | The dilution streaks of Novablue™ Gibson transformations were diluted again. |
| − | <b>18 Marie Eva</b> pSB1K3_PMnt_rfp was transformed. A colony PCR was run on Novablue™ | + | <b>18 Marie Eva</b> pSB1K3_PMnt_rfp was transformed. A colony PCR was run on |
| − | Gibson Assembly colonies. Gel electrophoresis was negative. <b>19 Marie Antonia</b> | + | Novablue™ |
| − | The colony PCR was repeated but remained negative. DH5-Alpha cells were transformed | + | Gibson Assembly colonies. Gel electrophoresis was negative. <b>19 Marie |
| | + | Antonia</b> |
| | + | The colony PCR was repeated but remained negative. DH5-Alpha cells were |
| | + | transformed |
| | with pMiniT2.0_cpp (tat, yta2, tp10, penetratin). A miniprep of pET-28b(+) was | | with pMiniT2.0_cpp (tat, yta2, tp10, penetratin). A miniprep of pET-28b(+) was |
| | performed. The product was controlled by gel electrophoresis but was negative | | performed. The product was controlled by gel electrophoresis but was negative |
| − | (Fig. CPP 3). <b>20 Marie Antonia</b> The BBa_R0073(Mnt)_rfp plasmid was digested to | + | (Fig. CPP 3). <b>20 Marie Antonia</b> The BBa_R0073(Mnt)_rfp plasmid was |
| | + | digested to |
| | isolate PMn_rfp and the backbone pSB1K3 was digested with respective enzymes. | | isolate PMn_rfp and the backbone pSB1K3 was digested with respective enzymes. |
| | pSB1K3_PMnt_rfp was ligated. synb1 was amplified by a PCR. A colony PCR was | | pSB1K3_PMnt_rfp was ligated. synb1 was amplified by a PCR. A colony PCR was |
| Line 11,374: |
Line 11,505: |
| | class="figure-img img-fluid rounded" | | class="figure-img img-fluid rounded" |
| | alt="Figure 14"></a> | | alt="Figure 14"></a> |
| − | <figcaption style="color: #F4F0DE; font-size: small;">Fig. CPP3 09/19/19 Miniprep of pET-28b(+)_synb1. Gel loaded (from left to right): Ladder 1 kb, pET-28b(+)_synb1 1, pET-28b(+)_synb1 2, pET-28b(+)_synb1 3.</figcaption> | + | <figcaption style="color: #F4F0DE; font-size: small;">Figure CPP3: 09/19/19 |
| | + | Miniprep of pET-28b(+)_synb1. Gel loaded (from left to right): Ladder 1 kb, |
| | + | pET-28b(+)_synb1 1, pET-28b(+)_synb1 2, pET-28b(+)_synb1 3. |
| | + | </figcaption> |
| | </figure> | | </figure> |
| | </div> | | </div> |
| Line 11,381: |
Line 11,515: |
| | </div> | | </div> |
| | | | |
| − | <div id="accordion12">
| + | <div id="accordion12"> |
| | <div class="card"> | | <div class="card"> |
| | <div class="card-header" id="headingTwelve"> | | <div class="card-header" id="headingTwelve"> |
| Line 11,396: |
Line 11,530: |
| | <div class="card-body" style="background-color: #232323;"> | | <div class="card-body" style="background-color: #232323;"> |
| | <p style="color: #F4F0DE"> | | <p style="color: #F4F0DE"> |
| − | <b>23 Marie Antonia</b> Transformation of pSB1K3_PMnt_rfp. The over-weekend cultures | + | <b>23 Marie Antonia</b> Transformation of pSB1K3_PMnt_rfp. The over-weekend |
| − | of Gibson assembly were used for a new colony PCR, lysing the cells before the reaction. | + | cultures |
| | + | of Gibson assembly were used for a new colony PCR, lysing the cells before the |
| | + | reaction. |
| | It remained negative. pET-28b(+) vector was digested and ligated with synb1. | | It remained negative. pET-28b(+) vector was digested and ligated with synb1. |
| − | The pET-28b(+)_synb1 was transformed. <b>24 Marie Eva</b> Overnight cultures of pSB1K3_PMnt_rfp | + | The pET-28b(+)_synb1 was transformed. <b>24 Marie Eva</b> Overnight cultures of |
| − | colonies were inoculated. Colony-PCR of pMiniT2.0_cpp was performed, gel electrophoresis | + | pSB1K3_PMnt_rfp |
| − | showed double bands (Fig. CPP 4). Overnight a ligation of pET-28b(+) and synb1 was performed. | + | colonies were inoculated. Colony-PCR of pMiniT2.0_cpp was performed, gel |
| − | <b>25 Marie Antonia</b> Nanodrop measuring of Gibson-PCR product. Transformation of pET-28b(+)_synb1. | + | electrophoresis |
| − | After Miniprep of pSB1K3_PMnt_rfp the plasmids were digested. Gel electrophoresis suggested | + | showed double bands (Fig. CPP 4). Overnight a ligation of pET-28b(+) and synb1 |
| − | ligation of the empty backbone. Single digestion of pSB1C3 and BBa_K091001 (K5) followed by | + | was performed. |
| − | Gibson assembly was repeated and DH5-Alpha cells transformed. <b>27 Antonia Marie</b> No growth of | + | <b>25 Marie Antonia</b> Nanodrop measuring of Gibson-PCR product. Transformation |
| − | new Gibson assembly. Over-weekend (RT) cultures of Novablue™ Gibson Assembly colonies were | + | of pET-28b(+)_synb1. |
| − | inoculated. Cultures of pMiniT2.0_penetratin and pET-28b(+)_synb1 colonies were inoculated. | + | After Miniprep of pSB1K3_PMnt_rfp the plasmids were digested. Gel |
| | + | electrophoresis suggested |
| | + | ligation of the empty backbone. Single digestion of pSB1C3 and BBa_K091001 (K5) |
| | + | followed by |
| | + | Gibson assembly was repeated and DH5-Alpha cells transformed. <b>27 Antonia |
| | + | Marie</b> No growth of |
| | + | new Gibson assembly. Over-weekend (RT) cultures of Novablue™ Gibson Assembly |
| | + | colonies were |
| | + | inoculated. Cultures of pMiniT2.0_penetratin and pET-28b(+)_synb1 colonies were |
| | + | inoculated. |
| | </p> | | </p> |
| | <figure> | | <figure> |
| Line 11,417: |
Line 11,562: |
| | class="figure-img img-fluid rounded" | | class="figure-img img-fluid rounded" |
| | alt="Figure 14"></a> | | alt="Figure 14"></a> |
| − | <figcaption style="color: #F4F0DE; font-size: small;">Fig. CPP 4. 09/24/19 Gel electrophoresis of Colony PCR. | + | <figcaption style="color: #F4F0DE; font-size: small;">Figure CPP 4: 09/24/19 Gel |
| − | Gel loaded (from left to right): Ladder 1 kb, penetratin 1, penetratin 2, tat 1, | + | electrophoresis of Colony PCR. |
| − | tat 2, YTA-2 1, YTA-2 2, tp10 1, TP 10 2. Bands expected at ~100 bp.</figcaption> | + | Gel loaded (from left to right): Ladder 1 kb, penetratin 1, penetratin 2, |
| | + | tat 1, |
| | + | tat 2, YTA-2 1, YTA-2 2, tp10 1, TP 10 2. Bands expected at ~100 bp. |
| | + | </figcaption> |
| | </figure> | | </figure> |
| | </div> | | </div> |
| Line 11,426: |
Line 11,574: |
| | </div> | | </div> |
| | | | |
| − | <div id="accordion13">
| + | <div id="accordion13"> |
| | <div class="card"> | | <div class="card"> |
| | <div class="card-header" id="headingThirteen"> | | <div class="card-header" id="headingThirteen"> |
| Line 11,441: |
Line 11,589: |
| | <div class="card-body" style="background-color: #232323;"> | | <div class="card-body" style="background-color: #232323;"> |
| | <p style="color: #F4F0DE"> | | <p style="color: #F4F0DE"> |
| − | <b>30 Marie Lina Antonia</b> The BBa_R0073(Mnt)_rfp plasmid was digested to isolate | + | <b>30 Marie Lina Antonia</b> The BBa_R0073(Mnt)_rfp plasmid was digested to |
| | + | isolate |
| | PMnt_rfp and the backbone pSB1K3 was digested with respective enzymes. | | PMnt_rfp and the backbone pSB1K3 was digested with respective enzymes. |
| | pSB1K3_PMnt_rfp was ligated. Miniprep of Novablue™ Gibson Assembly | | pSB1K3_PMnt_rfp was ligated. Miniprep of Novablue™ Gibson Assembly |
| Line 11,448: |
Line 11,597: |
| | inoculation of K2 (BBa_K608351) on plate. Repeating of colony-PCR of | | inoculation of K2 (BBa_K608351) on plate. Repeating of colony-PCR of |
| | pMiniT2.0_penetratin, pMiniT2.0_tat, pMiniT2.0_yta2, pMiniT2.0_tp10 colonies. | | pMiniT2.0_penetratin, pMiniT2.0_tat, pMiniT2.0_yta2, pMiniT2.0_tp10 colonies. |
| − | <b>01 Marie Lina</b> Antonia Digestion of Gibson assembly Minipreps and gel electrophoresis, | + | <b>01 Marie Lina</b> Antonia Digestion of Gibson assembly Minipreps and gel |
| | + | electrophoresis, |
| | which was negative (Fig. 15). Overnight cultures for K2 (BBa_K608351) | | which was negative (Fig. 15). Overnight cultures for K2 (BBa_K608351) |
| | were inoculated. Newly synthesized cpp+gfp parts arrived. Amplification | | were inoculated. Newly synthesized cpp+gfp parts arrived. Amplification |
| Line 11,456: |
Line 11,606: |
| | pSB1C3 and the constructs (K0-K15) were amplified via PCR (Taq polymerase). | | pSB1C3 and the constructs (K0-K15) were amplified via PCR (Taq polymerase). |
| | BBa_K575011 (Ev1), BBa_K523001 (Ev2), BBa_K763001 (Ev3), BBa_K763002 (Ev4) and | | BBa_K575011 (Ev1), BBa_K523001 (Ev2), BBa_K763001 (Ev3), BBa_K763002 (Ev4) and |
| − | BBa_K398108 (Ev5) from the iGEM distribution kit were transformed in DH5-Alpha cells. | + | BBa_K398108 (Ev5) from the iGEM distribution kit were transformed in DH5-Alpha |
| | + | cells. |
| | Amplification of CPP PM r10 (~100 bp) (Fig. CPP 6). Digest of CPPs. (Fig. CPP6). | | Amplification of CPP PM r10 (~100 bp) (Fig. CPP 6). Digest of CPPs. (Fig. CPP6). |
| | Retransformation of pET-28b(+). <b>04 Jakob Lina Antonia</b> Double Digestion of | | Retransformation of pET-28b(+). <b>04 Jakob Lina Antonia</b> Double Digestion of |
| Line 11,474: |
Line 11,625: |
| | class="figure-img img-fluid rounded" | | class="figure-img img-fluid rounded" |
| | alt="Figure 15"></a> | | alt="Figure 15"></a> |
| − | <figcaption style="color: #F4F0DE; font-size: small;">Fig. 15. 10/01/19 Digestion after Miniprep | + | <figcaption style="color: #F4F0DE; font-size: small;">Figure 15: 10/01/19 |
| − | (Gibson Assembly). Gel loaded (from left to right): Ladder 1 kb, pSB1C3_K1_K15 1-2, | + | Digestion after Miniprep |
| − | pSB1C3_K2_K3 1-2, K5_K4 1-2, pSB1C3_K6_K7 1-2, empty, empty, Miniprep | + | (Gibson Assembly). Gel loaded (from left to right): Ladder 1 kb, |
| | + | pSB1C3_K1_K15 1-2, |
| | + | pSB1C3_K2_K3 1-2, K5_K4 1-2, pSB1C3_K6_K7 1-2, empty, empty, Miniprep |
| | pSB1C3_K6_K7 undigested. | | pSB1C3_K6_K7 undigested. |
| | </figcaption> | | </figcaption> |
| Line 11,488: |
Line 11,641: |
| | class="figure-img img-fluid rounded" | | class="figure-img img-fluid rounded" |
| | alt="Figure 15"></a> | | alt="Figure 15"></a> |
| − | <figcaption style="color: #F4F0DE; font-size: small;">Fig. CPP 5. 10/01/19 PCR of CPP parts. Gel loaded (from left to right): Ladder 1 kb, | + | <figcaption style="color: #F4F0DE; font-size: small;">Figure CPP 5: 10/01/19 PCR |
| − | tp10+gfp, penetratin+gfp, yta2+gfp, pVec+gfp, synb1+gfp. All expected at 900kb. | + | of CPP parts. Gel loaded (from left to right): Ladder 1 kb, |
| | + | tp10+gfp, penetratin+gfp, yta2+gfp, pVec+gfp, synb1+gfp. All expected at |
| | + | 900kb. |
| | </figcaption> | | </figcaption> |
| | </figure> | | </figure> |
| Line 11,501: |
Line 11,656: |
| | class="figure-img img-fluid rounded" | | class="figure-img img-fluid rounded" |
| | alt="Figure 15"></a> | | alt="Figure 15"></a> |
| − | <figcaption style="color: #F4F0DE; font-size: small;">Fig. CPP 6. 10/02/19 Amplification of PM r10 with XbaI primer. (expected at 100 bp). Gel loaded (from left to right): r10 1.0, r10 2.0, r10 3.0. | + | <figcaption style="color: #F4F0DE; font-size: small;">Figure CPP 6: 10/02/19 |
| | + | Amplification of PM r10 with XbaI primer. (expected at 100 bp). Gel loaded |
| | + | (from left to right): r10 1.0, r10 2.0, r10 3.0. |
| | </figcaption> | | </figcaption> |
| | </figure> | | </figure> |
| Line 11,513: |
Line 11,670: |
| | class="figure-img img-fluid rounded" | | class="figure-img img-fluid rounded" |
| | alt="Figure 15"></a> | | alt="Figure 15"></a> |
| − | <figcaption style="color: #F4F0DE; font-size: small;">Fig. CPP 7. 10/02/19 Digest (expected at 100 bp). Gel | + | <figcaption style="color: #F4F0DE; font-size: small;">Fig. CPP 7. 10/02/19 |
| | + | Digest (expected at 100 bp). Gel |
| | loaded (from left to right): penetratin+gfp, penetratin+gfp (clean up), | | loaded (from left to right): penetratin+gfp, penetratin+gfp (clean up), |
| | tp10+gfp, tp10+gfp (clean up), pVec+gfp, pVec+gfp (clean up), synb1+gfp, | | tp10+gfp, tp10+gfp (clean up), pVec+gfp, pVec+gfp (clean up), synb1+gfp, |
| Line 11,525: |
Line 11,683: |
| | </div> | | </div> |
| | | | |
| − | <div id="accordion14">
| + | <div id="accordion14"> |
| | <div class="card"> | | <div class="card"> |
| | <div class="card-header" id="headingFourteen"> | | <div class="card-header" id="headingFourteen"> |
| Line 11,540: |
Line 11,698: |
| | <div class="card-body" style="background-color: #232323;"> | | <div class="card-body" style="background-color: #232323;"> |
| | <p style="color: #F4F0DE"> | | <p style="color: #F4F0DE"> |
| − | <b>07 Jakob Lina Antonia</b> Overnight cultures of pSB1C3_K0_K15 (Gibson), K5_K4(Gibson), | + | <b>07 Jakob Lina Antonia</b> Overnight cultures of pSB1C3_K0_K15 (Gibson), |
| | + | K5_K4(Gibson), |
| | K5_K4 and pSB1C3_K7 were inoculated. Miniprep of Ev1-Ev5 over-weekend cultures. | | K5_K4 and pSB1C3_K7 were inoculated. Miniprep of Ev1-Ev5 over-weekend cultures. |
| | Transformation of BBa_K132018, BBa_K098991, BBa_K876004 and BBa_K74100X from | | Transformation of BBa_K132018, BBa_K098991, BBa_K876004 and BBa_K74100X from |
| − | iGEM distribution kit. <b>08 Antonia</b> Miniprep of construct overnight cultures was | + | iGEM distribution kit. <b>08 Antonia</b> Miniprep of construct overnight |
| | + | cultures was |
| | conducted, followed by single digest, which suggested unsuccessful ligations. | | conducted, followed by single digest, which suggested unsuccessful ligations. |
| − | <b>09 Lina</b> Digest of PM r10 CPP and pET-28b(+) (Fig. CPP 8). <b>12 Steffen Famke Antonia</b> | + | <b>09 Lina</b> Digest of PM r10 CPP and pET-28b(+) (Fig. CPP 8). <b>12 Steffen |
| − | The characterisation of BBa_K398108 (Ev5), a salt tolerant cluster, was proceeded | + | Famke Antonia</b> |
| − | after identity of the plasmid was confirmed via digestion (Fig. 16). An overnight | + | The characterisation of BBa_K398108 (Ev5), a salt tolerant cluster, was |
| − | culture of the bacteria (from which the Ev5 mini originated), as well as DH5alpha | + | proceeded |
| − | wild type cells were inoculated in LB and put on the shaker at 37°C. As a basis | + | after identity of the plasmid was confirmed via digestion (Fig. 16). An |
| − | for our modeling, the growth of <i>E. coli</i> Nissle 1917 under in minimal media was to | + | overnight |
| − | be analysed. For this, an overnight culture was inoculated and M9 medium was prepared. | + | culture of the bacteria (from which the Ev5 mini originated), as well as |
| − | <b>13 Lina Jakob Famke Antonia</b> The salt tolerance experiment was performed. For this | + | DH5alpha |
| − | LB 0M KCl, 0.05M KCl, 0.1M KCl, 0.2M KCl, 0.4M KCl and 0.8M KCl in LB were prepared | + | wild type cells were inoculated in LB and put on the shaker at 37°C. As a basis |
| | + | for our modeling, the growth of <i>E. coli</i> Nissle 1917 under in minimal |
| | + | media was to |
| | + | be analysed. For this, an overnight culture was inoculated and M9 medium was |
| | + | prepared. |
| | + | <b>13 Lina Jakob Famke Antonia</b> The salt tolerance experiment was performed. |
| | + | For this |
| | + | LB 0M KCl, 0.05M KCl, 0.1M KCl, 0.2M KCl, 0.4M KCl and 0.8M KCl in LB were |
| | + | prepared |
| | and sterile filtered. The experiment was then performed in triplicates with | | and sterile filtered. The experiment was then performed in triplicates with |
| | each DH5alpha wild type and the salt cluster bearing DH5alpha cells. Of each | | each DH5alpha wild type and the salt cluster bearing DH5alpha cells. Of each |
| Line 11,559: |
Line 11,727: |
| | every 45min using the Nanophotometer of Implen. The experiment was finished | | every 45min using the Nanophotometer of Implen. The experiment was finished |
| | upon reaching the end of the exponential phase. The measured data (Fig. 17), | | upon reaching the end of the exponential phase. The measured data (Fig. 17), |
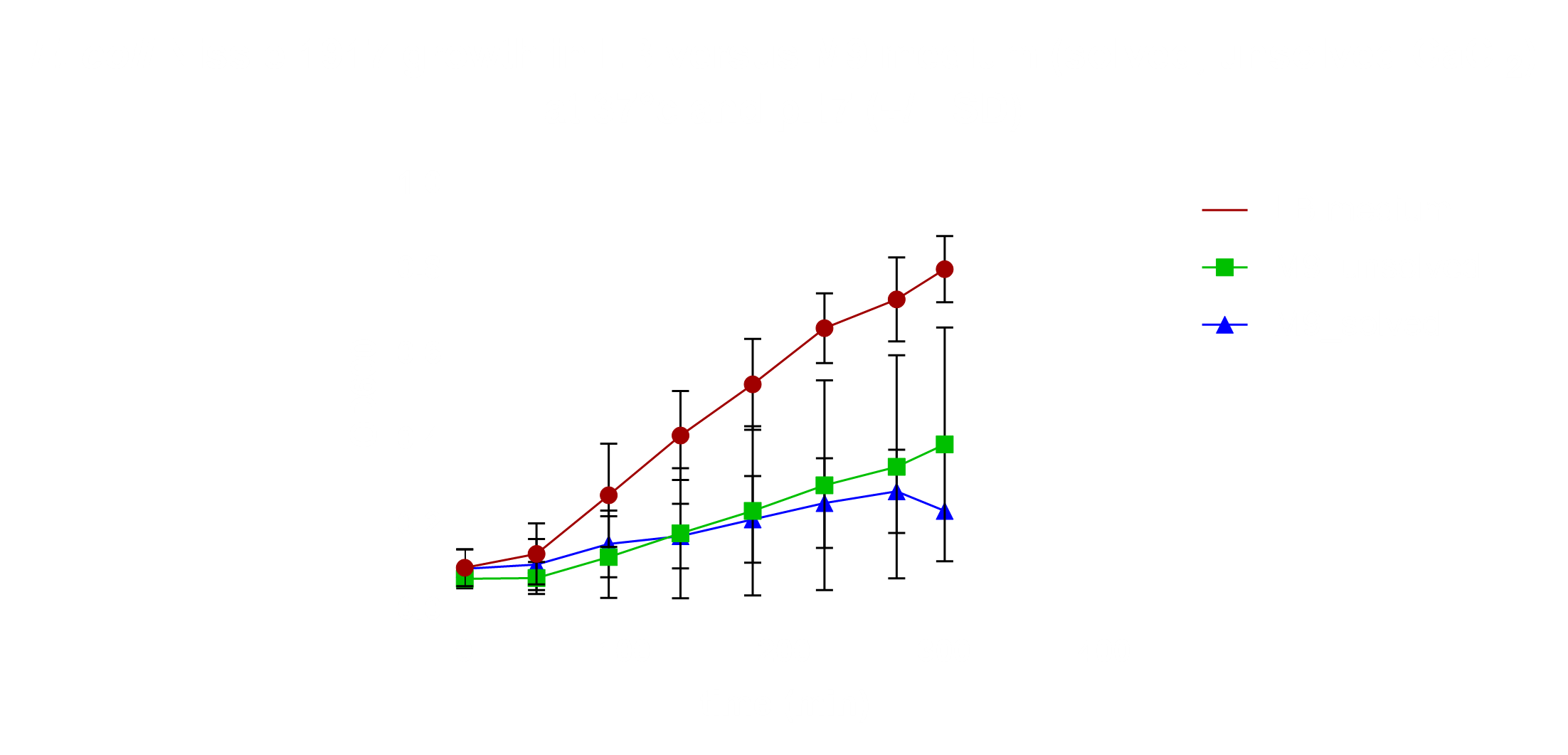
| − | was logarithmized, nonlinearly fitted and normalised against each other (Fig. 18), | + | was logarithmized, nonlinearly fitted and normalised against each other (Fig. |
| | + | 18), |
| | yielding the result that the salt tolerance cluster does not confer to increased | | yielding the result that the salt tolerance cluster does not confer to increased |
| − | tolerance against KCl in <i>E. coli</i> DH5alpha. The minimal medium M9 was inoculated | + | tolerance against KCl in <i>E. coli</i> DH5alpha. The minimal medium M9 was |
| | + | inoculated |
| | with EcN overnight cultures. Since calcium chloride did not dissolve properly, | | with EcN overnight cultures. Since calcium chloride did not dissolve properly, |
| | one group of four samples in M9 was used with the unsolved. After several hours | | one group of four samples in M9 was used with the unsolved. After several hours |
| Line 11,572: |
Line 11,742: |
| | <p> | | <p> |
| | Please find the recipe for the M9 medium provided by Arie Geerlof - | | Please find the recipe for the M9 medium provided by Arie Geerlof - |
| − | Helmholtz Center Munich under the following link: <a href="https://static.igem.org/mediawiki/2019/2/20/T--Tuebingen--M9_recipe.pdf" onclick='window.open("https://static.igem.org/mediawiki/2019/2/20/T--Tuebingen--M9_recipe.pdf","_self");'>https://static.igem.org/mediawiki/2019/2/20/T--Tuebingen--M9_recipe.pdf</a> | + | Helmholtz Center Munich under the following link: <a |
| | + | href="https://static.igem.org/mediawiki/2019/2/20/T--Tuebingen--M9_recipe.pdf" |
| | + | onclick='window.open("https://static.igem.org/mediawiki/2019/2/20/T--Tuebingen--M9_recipe.pdf","_self");'>https://static.igem.org/mediawiki/2019/2/20/T--Tuebingen--M9_recipe.pdf</a> |
| | </p> | | </p> |
| | | | |
| Line 11,583: |
Line 11,755: |
| | class="figure-img img-fluid rounded" | | class="figure-img img-fluid rounded" |
| | alt="Figure 16"></a> | | alt="Figure 16"></a> |
| − | <figcaption style="color: #F4F0DE; font-size: small;">Fig. CPP 8. 10/09/19 Digest of PM r10 CPP and pET-28b(+). Gel loaded (from left to right): PM r10 CPP, PM r10 CPP, pET-28b(+), pET-28b(+), pET-28b(+) NcoI, pET-28b(+) EcoRI </figcaption> | + | <figcaption style="color: #F4F0DE; font-size: small;">Figure CPP 8: 10/09/19 |
| | + | Digest of PM r10 CPP and pET-28b(+). Gel loaded (from left to right): PM r10 |
| | + | CPP, PM r10 CPP, pET-28b(+), pET-28b(+), pET-28b(+) NcoI, pET-28b(+) EcoRI |
| | + | </figcaption> |
| | </figure> | | </figure> |
| | | | |
| | <div class="row align-items-center"> | | <div class="row align-items-center"> |
| − | <div class="col-12 col-md-9">
| + | <div class="col-12 col-md-9"> |
| − | <figure>
| + | <figure> |
| − | <a style="font-size: small"
| + | <a style="font-size: small" |
| − | href="https://static.igem.org/mediawiki/2019/3/3d/T--Tuebingen--Gel16.png"
| + | href="https://static.igem.org/mediawiki/2019/3/3d/T--Tuebingen--Gel16.png" |
| − | data-effect="mfp-zoom-out"
| + | data-effect="mfp-zoom-out" |
| − | title="Figure 16">
| + | title="Figure 16"> |
| − | <img src="https://static.igem.org/mediawiki/2019/3/3d/T--Tuebingen--Gel16.png"
| + | <img src="https://static.igem.org/mediawiki/2019/3/3d/T--Tuebingen--Gel16.png" |
| − | class="figure-img img-fluid rounded"
| + | class="figure-img img-fluid rounded" |
| − | alt="Figure 16"></a>
| + | alt="Figure 16"></a> |
| − | <figcaption style="color: #F4F0DE; font-size: small;">Fig. 16. 10/12/19 Digestion after Miniprep. Gel loaded (from left to right): Ladder 1 kb, BBa_K575011 (Ev1) single digest, BBa_K575011 (Ev1) double digest, BBa_K763002 (Ev4) single digest, BBa_K763002 (Ev4) double digest, BBa_K398108 (Ev5) single digest, BBa_K398108 (Ev5) double digest.</figcaption>
| + | <figcaption style="color: #F4F0DE; font-size: small;">Figure 16: |
| − | </figure>
| + | 10/12/19 Digestion after Miniprep. Gel loaded (from left to right): |
| − | </div>
| + | Ladder 1 kb, BBa_K575011 (Ev1) single digest, BBa_K575011 (Ev1) |
| | + | double digest, BBa_K763002 (Ev4) single digest, BBa_K763002 (Ev4) |
| | + | double digest, BBa_K398108 (Ev5) single digest, BBa_K398108 (Ev5) |
| | + | double digest. |
| | + | </figcaption> |
| | + | </figure> |
| | + | </div> |
| | </div> | | </div> |
| | | | |
| | <div class="row align-items-center"> | | <div class="row align-items-center"> |
| − | <div class="col-12 col-md-9">
| + | <div class="col-12 col-md-9"> |
| − | <figure>
| + | <figure> |
| − | <a style="font-size: small"
| + | <a style="font-size: small" |
| − | href="https://static.igem.org/mediawiki/2019/e/eb/T--Tuebingen--figure17-white.png"
| + | href="https://static.igem.org/mediawiki/2019/e/eb/T--Tuebingen--figure17-white.png" |
| − | data-effect="mfp-zoom-out"
| + | data-effect="mfp-zoom-out" |
| − | title="Fig. 17. Mean of the triplicates and standard deviation of the measured growth curves. ">
| + | title="Fig. 17. Mean of the triplicates and standard deviation of the measured growth curves. "> |
| − | <img src="https://static.igem.org/mediawiki/2019/e/eb/T--Tuebingen--figure17-white.png"
| + | <img src="https://static.igem.org/mediawiki/2019/e/eb/T--Tuebingen--figure17-white.png" |
| − | class="figure-img img-fluid rounded"
| + | class="figure-img img-fluid rounded" |
| − | alt="Figure 16"></a>
| + | alt="Figure 16"></a> |
| − | <figcaption style="color: #F4F0DE; font-size: small;">Fig. 17. Mean of the triplicates and standard deviation of the measured growth curves. </figcaption>
| + | <figcaption style="color: #F4F0DE; font-size: small;">Figure 17: Mean of |
| − | </figure>
| + | the triplicates and standard deviation of the measured growth |
| − | </div>
| + | curves. |
| | + | </figcaption> |
| | + | </figure> |
| | + | </div> |
| | </div> | | </div> |
| | <figure> | | <figure> |
| Line 11,623: |
Line 11,807: |
| | class="figure-img img-fluid rounded" | | class="figure-img img-fluid rounded" |
| | alt="Figure 16"></a> | | alt="Figure 16"></a> |
| − | <figcaption style="color: #F4F0DE; font-size: small;">Fig. 18. Growth rate [h-1] and [%] growth of tolerant normalised to wt <i>E. coli</i> DH5alpha.Fit was performed with the mean of the three samples.</figcaption> | + | <figcaption style="color: #F4F0DE; font-size: small;">Figure 18: Growth rate |
| | + | [h-1] and [%] growth of tolerant normalised to wt <i>E. coli</i> |
| | + | DH5alpha.Fit was performed with the mean of the three samples. |
| | + | </figcaption> |
| | </figure> | | </figure> |
| | | | |
| Line 11,634: |
Line 11,821: |
| | class="figure-img img-fluid rounded" | | class="figure-img img-fluid rounded" |
| | alt="Figure 16"></a> | | alt="Figure 16"></a> |
| − | <figcaption style="color: #F4F0DE; font-size: small;">Fig. 19. Comparison of EcN growth in minimal medium | + | <figcaption style="color: #F4F0DE; font-size: small;">Figure 19: Comparison of |
| − | (M9 solved/unsolved CaCl2) versus LB medium at pH7 and 37°C. </figcaption> | + | EcN growth in minimal medium |
| | + | (M9 solved/unsolved CaCl2) versus LB medium at pH7 and 37°C. |
| | + | </figcaption> |
| | </figure> | | </figure> |
| | | | |
| − | <h3 style="font-family: Righteous; color: #2ecc71">Rna_Seq</h3> | + | <h3 style="font-family: Righteous; color: #2ecc71">RNA-Seq</h3> |
| | <p> | | <p> |
| − | For the preparation of the cultures of <i>E. coli</i> Nissle 1917 Lina used LB-Lennox medium. For one sample the pH was adjusted to pH 4 and was incubated at 37°C. In another sample 100 µM H2O2 were added and incubated at 37°C. The remaining three samples were incubated at different temperatures (8,25,37°C). | + | For the preparation of the cultures of <i>E. coli</i> Nissle 1917 Lina used |
| | + | LB-Lennox medium. For one sample the pH was adjusted to pH 4 and was incubated |
| | + | at 37°C. In another sample 100 µM H2O2 were added and incubated at 37°C. The |
| | + | remaining three samples were incubated at different temperatures (8,25,37°C). |
| | </p> | | </p> |
| | <p> | | <p> |
| − | After pelleting the bacterial cultures the pellet was taken up in 1 ml RNAlater to ensure that the RNA is not degraded and expression levels do not change. Patrick and Lina isolated the RNA from the 24 samples with the PureLink RNA Mini Kit by Thermo Fisher Scientific using 500ml of the diluted pellet. The isolated nucleic acid was checked via an agarose gel and the amount estimated via Qubit measurement before being treated with DNase I by Roche. | + | After pelleting the bacterial cultures the pellet was taken up in 1 ml RNAlater |
| | + | to ensure that the RNA is not degraded and expression levels do not change. |
| | + | Patrick and Lina isolated the RNA from the 24 samples with the PureLink RNA Mini |
| | + | Kit by Thermo Fisher Scientific using 500ml of the diluted pellet. The isolated |
| | + | nucleic acid was checked via an agarose gel and the amount estimated via Qubit |
| | + | measurement before being treated with DNase I by Roche. |
| | </p> | | </p> |
| | <div class="row align-items-center"> | | <div class="row align-items-center"> |
| − | <div class="col-12 col-md-5">
| |
| − | <figure>
| |
| − | <a style="font-size: small"
| |
| − | href="https://static.igem.org/mediawiki/2019/5/58/T--Tuebingen--NCCT-RNA1_PNG.png"
| |
| − | data-effect="mfp-zoom-out"
| |
| − | title="Fig.20:Gel electrophoresis of the RNA after RNA isolation of each sample.">
| |
| − | <img src="https://static.igem.org/mediawiki/2019/5/58/T--Tuebingen--NCCT-RNA1_PNG.png"
| |
| − | class="figure-img img-fluid rounded"
| |
| − | alt="Placeholder"></a>
| |
| − | <figcaption style="color: #F4F0DE; font-size: small;">Fig.20:Gel electrophoresis of the RNA after RNA isolation of each sample. From left to right: 1 kb Ladder, 4x LB aerobe 37°C, 4x LB aerobe 25°C, 4x LB aerobe 8°C, 1 kb Ladder, 4x LB aerobe H2O2, 4x LB aerobe pH4, 4x LB aerobe dry freeze. The two strong bands in each column represent the rRNA which was depleted in further steps.</figcaption>
| |
| − | </figure>
| |
| − |
| |
| − | </div>
| |
| − | <div class="col-12 col-md-5">
| |
| | <figure> | | <figure> |
| − | <a style="font-size: small"
| + | <a style="font-size: small" |
| − | href="https://static.igem.org/mediawiki/2019/6/6d/T--Tuebingen--NCCT-RNA-2.png"
| + | href="https://static.igem.org/mediawiki/2019/5/51/T--Tuebingen--NCCT-RNA-neu.png" |
| − | data-effect="mfp-zoom-out"
| + | data-effect="mfp-zoom-out" |
| − | title="Fig.20:Gel electrophoresis of the RNA after RNA isolation of each sample.">
| + | title="Fig.20:Gel electrophoresis of the RNA after RNA isolation of each sample."> |
| − | <img src="https://static.igem.org/mediawiki/2019/6/6d/T--Tuebingen--NCCT-RNA-2.png"
| + | <img src="https://static.igem.org/mediawiki/2019/5/51/T--Tuebingen--NCCT-RNA-neu.png" |
| − | class="figure-img img-fluid rounded"
| + | class="figure-img img-fluid rounded" |
| − | alt="Placeholder"></a>
| + | alt="Placeholder"></a> |
| − | <figcaption style="color: #F4F0DE; font-size: small;">Fig.20:Gel electrophoresis of the RNA after RNA isolation of each sample. From left to right: 1 kb Ladder, 4x LB aerobe 37°C, 4x LB aerobe 25°C, 4x LB aerobe 8°C, 1 kb Ladder, 4x LB aerobe H2O2, 4x LB aerobe pH4, 4x LB aerobe dry freeze. The two strong bands in each column represent the rRNA which was depleted in further steps.</figcaption> | + | <figcaption style="color: #F4F0DE; font-size: small;">Figure 20: Gel |
| − | </figure>
| + | electrophoresis of the RNA after RNA isolation of each sample. From left |
| − | </div>
| + | to right: 1 kb Ladder, 4x LB aerobe 37°C, 4x LB aerobe 25°C, 4x LB |
| − | </div>
| + | aerobe 8°C, 1 kb Ladder, 4x LB aerobe H2O2, 4x LB aerobe pH4, 4x LB |
| | + | aerobe dry freeze. The two strong bands in each column represent the |
| | + | rRNA which was depleted in further steps. |
| | + | </figcaption> |
| | + | </figure> |
| | + | |
| | + | </div> |
| | <p> | | <p> |
| − | Patrick purified the RNA via the RNA Clean & Concentrator Kit by Zymo. Another agarose gel was conducted and RNA amount estimated via Qubit. rRNA depletion was conducted with the riboPOOL kit by siTOOLs, the RNA was again purified with the RNA Clean & Concentrator Kit by Zymo and RNA amount estimated via Qubit measurement. Despite very low RNA amount it was proceeded with the Library preparation with the CORALL Total RNA-Seq Library Prep Kit by Lexogen. RNA amounts were analysed via Qubit measurement and Library quality was analysed via FemtoPulse measurement by Agilent. Additionally a qPCR of a pool of all 24 libraries was conducted to better determine the Library amount with the NEBNext Library Quant Kit by NEB. Since the library quality was not sufficient, the rest of the DNase I treated samples was used to start depletion and library preparation again. This time the Total RNA Seq Library Prep Kit by Zymo was used, RNA amount estimated via Qubit measurement and Library quality analysed via FemtoPulse as well as a qPCR. Again RNA amount was low after depletion and library preparation and library quality was not high enough for sequencing. Jakob repeated the <i>E. coli</i> growth and the RNA Extraction with the Qiagen RNeasy kit instead of the dry freezed samples the condition was changed to LB aerob with pH8. The nucleic acid concentration was high after extraction and the RNA showed no sign of degradation. | + | Patrick purified the RNA via the RNA Clean & Concentrator Kit by Zymo. Another |
| | + | agarose gel was conducted and RNA amount estimated via Qubit. rRNA depletion was |
| | + | conducted with the riboPOOL kit by siTOOLs, the RNA was again purified with the |
| | + | RNA Clean & Concentrator Kit by Zymo and RNA amount estimated via Qubit |
| | + | measurement. Despite very low RNA amount it was proceeded with the Library |
| | + | preparation with the CORALL Total RNA-Seq Library Prep Kit by Lexogen. RNA |
| | + | amounts were analysed via Qubit measurement and Library quality was analysed via |
| | + | FemtoPulse measurement by Agilent. Additionally a qPCR of a pool of all 24 |
| | + | libraries was conducted to better determine the Library amount with the NEBNext |
| | + | Library Quant Kit by NEB. Since the library quality was not sufficient, the rest |
| | + | of the DNase I treated samples was used to start depletion and library |
| | + | preparation again. This time the Total RNA Seq Library Prep Kit by Zymo was |
| | + | used, RNA amount estimated via Qubit measurement and Library quality analysed |
| | + | via FemtoPulse as well as a qPCR. Again RNA amount was low after depletion and |
| | + | library preparation and library quality was not high enough for sequencing. |
| | + | Jakob repeated the <i>E. coli</i> growth and the RNA Extraction with the Qiagen |
| | + | RNeasy kit instead of the dry freezed samples the condition was changed to LB |
| | + | aerob with pH8. The nucleic acid concentration was high after extraction and the |
| | + | RNA showed no sign of degradation. |
| | </p> | | </p> |
| | <p> | | <p> |
| − | A DNAse digestion was conducted, the cDNA synthesis, the library preparation and the rRNA depletion were done with the Universal Prokaryotic RNA-Seq Prokaryotic AnyDeplete ™ kit from NuGEN. At a first glance, the library preparation seemed successful. Soon, the pooled libraries will be loaded onto the Illumina MiSeq System for sequencing. | + | A DNAse digestion was conducted, the cDNA synthesis, the library preparation and |
| | + | the rRNA depletion were done with the Universal Prokaryotic RNA-Seq Prokaryotic |
| | + | AnyDeplete ™ kit from NuGEN. At a first glance, the library preparation seemed |
| | + | successful. Soon, the pooled libraries will be loaded onto the Illumina MiSeq |
| | + | System for sequencing. |
| | </p> | | </p> |
| | | | |
| − | <figure>
| + | <figure> |
| − | <a style="font-size: small"
| + | <a style="font-size: small" |
| − | href="https://static.igem.org/mediawiki/2019/4/45/T--Tuebingen--RNA-2-NCCT-jakob.png"
| + | href="https://static.igem.org/mediawiki/2019/4/45/T--Tuebingen--RNA-2-NCCT-jakob.png" |
| − | data-effect="mfp-zoom-out"
| + | data-effect="mfp-zoom-out" |
| − | title="Fig.21: Gel electrophoresis of the RNA after RNA isolation of each sample.">
| + | title="Fig.21: Gel electrophoresis of the RNA after RNA isolation of each sample."> |
| − | <img src="https://static.igem.org/mediawiki/2019/4/45/T--Tuebingen--RNA-2-NCCT-jakob.png"
| + | <img src="https://static.igem.org/mediawiki/2019/4/45/T--Tuebingen--RNA-2-NCCT-jakob.png" |
| − | class="figure-img img-fluid rounded"
| + | class="figure-img img-fluid rounded" |
| − | alt="Placeholder"></a>
| + | alt="Placeholder"></a> |
| − | <figcaption style="color: #F4F0DE; font-size: small;">Fig.21: Gel electrophoresis of the RNA after RNA isolation of each sample.
| + | <figcaption style="color: #F4F0DE; font-size: small;">Figure 21: Gel |
| − | From left to right: 1 kb Ladder, 1x RNA before cleanup 4x LB aerobe H2O2, 4x LB aerobe
| + | electrophoresis of the RNA after RNA isolation of each sample. |
| − | 8°C, 4x LB aerobe 25°C, 1x LB aerobe 37°C, 1 kb Ladder 1x RNA before cleanup 3x LB aerobe
| + | From left to right: 1 kb Ladder, 1x RNA before cleanup 4x LB aerobe H2O2, 4x |
| − | 37°C, 4x LB aerobe pH4, 4x Lb aerob pH8. The two strong bands in each column represent
| + | LB aerobe |
| − | the rRNA which was depleted in further steps</figcaption>
| + | 8°C, 4x LB aerobe 25°C, 1x LB aerobe 37°C, 1 kb Ladder 1x RNA before cleanup |
| − | </figure>
| + | 3x LB aerobe |
| | + | 37°C, 4x LB aerobe pH4, 4x Lb aerob pH8. The two strong bands in each column |
| | + | represent |
| | + | the rRNA which was depleted in further steps |
| | + | </figcaption> |
| | + | </figure> |
| | <p> | | <p> |
| − | Lina and Katharina prepared the anaerob bacterial cultures with the different conditions: LB aerob, Lb anaerob, mGAM medium anaerob, mGAM medium with 1.3 mM Metformin anaerob, mGAM medium with 0.25 mM cholic acid anaerob and mGAM medium with the supernatant of a Bacteroides thataiotaomicron culture. Cultures were again pelleted and taken up in RNAlater. Patrick isolated the RNA with the PureLink RNA Mini Kit by Thermo Fisher Scientific and conducted a Qubit measurement before and after treatment with DNaseI by Roche. Jakob and Steffen tested the quality of the RNA via an Agilent RNA 6000 Nano Bioanalyzer. Degradation was clearly visible but the samples could be used for the library prep. The rRNA was depleted via the NEBNext (R) rRNA Depletion Kit. Thereafter, our collaborators at the EMBL conducted the automated RNA library preparation and sequencing. | + | Lina and Katharina prepared the anaerob bacterial cultures with the different |
| | + | conditions: LB aerob, Lb anaerob, mGAM medium anaerob, mGAM medium with 1.3 mM |
| | + | Metformin anaerob, mGAM medium with 0.25 mM cholic acid anaerob and mGAM medium |
| | + | with the supernatant of a Bacteroides thataiotaomicron culture. Cultures were |
| | + | again pelleted and taken up in RNAlater. Patrick isolated the RNA with the |
| | + | PureLink RNA Mini Kit by Thermo Fisher Scientific and conducted a Qubit |
| | + | measurement before and after treatment with DNaseI by Roche. Jakob and Steffen |
| | + | tested the quality of the RNA via an Agilent RNA 6000 Nano Bioanalyzer. |
| | + | Degradation was clearly visible but the samples could be used for the library |
| | + | prep. The rRNA was depleted via the NEBNext (R) rRNA Depletion Kit. Thereafter, |
| | + | our collaborators at the EMBL conducted the automated RNA library preparation |
| | + | and sequencing. |
| | </p> | | </p> |
| | </div> | | </div> |
| Line 11,709: |
Line 11,936: |
| | | | |
| | | | |
| − | <script src="https://2019.igem.org/Team:Tuebingen/sidebarJS?action=raw&ctype=text/javascript"></script>
| + | <script src="https://2019.igem.org/Team:Tuebingen/sidebarJS?action=raw&ctype=text/javascript"></script> |
| | | | |
| − | <script src="https://2019.igem.org/Team:Tuebingen/smoothScrollJS?action=raw&ctype=text/javascript"></script>
| + | <script src="https://2019.igem.org/Team:Tuebingen/smoothScrollJS?action=raw&ctype=text/javascript"></script> |
| − | <script>
| + | <script> |
| − | var scroll = new SmoothScroll('a[href*="#"]');
| + | var scroll = new SmoothScroll('a[href*="#"]'); |
| − | </script>
| + | </script> |
| − | <script src="https://2019.igem.org/Team:Tuebingen/magni-popup-minJS?action=raw&ctype=text/javascript"></script>
| + | <script src="https://2019.igem.org/Team:Tuebingen/magni-popup-minJS?action=raw&ctype=text/javascript"></script> |
| − | <script src="https://2019.igem.org/Team:Tuebingen/popup-imagesJS?action=raw&ctype=text/javascript"></script>
| + | <script src="https://2019.igem.org/Team:Tuebingen/popup-imagesJS?action=raw&ctype=text/javascript"></script> |
| | | | |
| − | <div class='container-fluid p-3' style="background-color: #919191;">
| + | <div class='container-fluid p-3' style="background-color: #919191;"> |
| − | <hr>
| + | <hr> |
| − | <div class='row align-items-center'>
| + | <div class='row align-items-center'> |
| − | <div class='col-6 col-md-2 sponsorLogo text-center'>
| + | <div class='col-6 col-md-2 sponsorLogo text-center'> |
| − | <a href="https://www.stura-tuebingen.de/" target="_blank2"><img class="img-fluid"
| + | <a href="https://www.stura-tuebingen.de/" target="_blank2"><img class="img-fluid" |
| − | src="https://static.igem.org/mediawiki/2019/1/10/T--Tuebingen--stura_logo.png"
| + | src="https://static.igem.org/mediawiki/2019/1/10/T--Tuebingen--stura_logo.png" |
| − | style="max-height: 80px;"></a>
| + | style="max-height: 80px;"></a> |
| − | </div>
| + | </div> |
| − |
| + | |
| − | <div class='col-6 col-md-2 sponsorLogo text-center'>
| + | <div class='col-6 col-md-2 sponsorLogo text-center'> |
| − | <a href="https://www.kohlbacherlab.org" target="_blank2"><img class="img-fluid"
| + | <a href="https://www.kohlbacherlab.org" target="_blank2"><img class="img-fluid" |
| − | src="https://static.igem.org/mediawiki/2019/9/99/T--Tuebingen--kohlbacher_logo.png"
| + | src="https://static.igem.org/mediawiki/2019/9/99/T--Tuebingen--kohlbacher_logo.png" |
| − | style="max-height: 80px;"></a>
| + | style="max-height: 80px;"></a> |
| − | </div>
| + | </div> |
| − | <div class='col-6 col-md-2 sponsorLogo text-center'>
| + | <div class='col-6 col-md-2 sponsorLogo text-center'> |
| − | <a href="https://www.dieter-schwarz-stiftung.de/homepage.html" target="_blank2"><img class="img-fluid"
| + | <a href="https://www.dieter-schwarz-stiftung.de/homepage.html" target="_blank2"><img class="img-fluid" |
| − | src="https://static.igem.org/mediawiki/2019/8/86/T--Tuebingen--LogoDSS.png"
| + | src="https://static.igem.org/mediawiki/2019/8/86/T--Tuebingen--LogoDSS.png" |
| − | style="max-height: 80px;"></a>
| + | style="max-height: 80px;"></a> |
| − | </div>
| + | </div> |
| − |
| + | |
| − | <div class='col-6 col-md-2 sponsorLogo text-center'>
| + | <div class='col-6 col-md-2 sponsorLogo text-center'> |
| − | <a href="https://www.embl.de/" target="_blank2"><img class="img-fluid"
| + | <a href="https://www.embl.de/" target="_blank2"><img class="img-fluid" |
| − | src="https://static.igem.org/mediawiki/2019/c/c4/T--Tuebingen--embl_logo.png"
| + | src="https://static.igem.org/mediawiki/2019/c/c4/T--Tuebingen--embl_logo.png" |
| − | style="max-height: 80px;"></a>
| + | style="max-height: 80px;"></a> |
| − | </div>
| + | </div> |
| − |
| + | |
| − | <div class='col-6 col-md-2 sponsorLogo text-center'>
| + | <div class='col-6 col-md-2 sponsorLogo text-center'> |
| − | <a href="https://www.experimenta.science/en/" target="_blank2"><img class="img-fluid"
| + | <a href="https://www.experimenta.science/en/" target="_blank2"><img class="img-fluid" |
| − | src="https://static.igem.org/mediawiki/2019/e/eb/T--Tuebingen--Logo-Experimenta.png"
| + | src="https://static.igem.org/mediawiki/2019/e/eb/T--Tuebingen--Logo-Experimenta.png" |
| | + | style="max-height: 80px;"></a> |
| | + | </div> |
| | + | |
| | + | <div class='col-6 col-md-2 sponsorLogo text-center'> |
| | + | <a href="https://www.medizin.uni-tuebingen.de/de/medizinische-fakultaet/forschung/core-facilities/ncct" |
| | + | target="_blank2"><img class="img-fluid" |
| | + | src="https://static.igem.org/mediawiki/2019/e/ec/T--Tuebingen--NCCT.png" |
| | + | style="max-height: 80px;"></a> |
| | + | </div> |
| | + | |
| | + | </div> |
| | + | |
| | + | <hr> |
| | + | <div class='row align-items-center'> |
| | + | <div class='col-4 col-md-2 col-xl-1 sponsorLogo text-center'> |
| | + | <a href="https://www.lexogen.com/" target="_blank2"><img class="img-fluid" |
| | + | src="https://static.igem.org/mediawiki/2019/4/43/T--Tuebingen--lexogen_logo.png"></a> |
| | + | </div> |
| | + | <div class='col-4 col-md-2 col-xl-1 sponsorLogo text-center'> |
| | + | <a href="https://www.zmbp.uni-tuebingen.de/zmbp.html" target="_blank2"><img class="img-fluid" |
| | + | src="https://static.igem.org/mediawiki/2019/d/d8/T--Tuebingen--zmbp_logo.png" |
| | style="max-height: 80px;"></a> | | style="max-height: 80px;"></a> |
| − | </div>
| |
| − |
| |
| − | <div class='col-6 col-md-2 sponsorLogo text-center'>
| |
| − | <a href="https://www.medizin.uni-tuebingen.de/de/medizinische-fakultaet/forschung/core-facilities/ncct"
| |
| − | target="_blank2"><img class="img-fluid"
| |
| − | src="https://static.igem.org/mediawiki/2019/e/ec/T--Tuebingen--NCCT.png"
| |
| − | style="max-height: 80px;"></a>
| |
| − | </div>
| |
| − |
| |
| − | </div>
| |
| − |
| |
| − | <hr>
| |
| − | <div class='row align-items-center'>
| |
| − | <div class='col-4 col-md-2 col-xl-1 sponsorLogo text-center'>
| |
| − | <a href="https://www.lexogen.com/" target="_blank2"><img class="img-fluid"
| |
| − | src="https://static.igem.org/mediawiki/2019/4/43/T--Tuebingen--lexogen_logo.png"></a>
| |
| − | </div>
| |
| − | <div class='col-4 col-md-2 col-xl-1 sponsorLogo text-center'>
| |
| − | <a href="https://www.zmbp.uni-tuebingen.de/zmbp.html" target="_blank2"><img class="img-fluid"
| |
| − | src="https://static.igem.org/mediawiki/2019/d/d8/T--Tuebingen--zmbp_logo.png"
| |
| − | style="max-height: 80px;"></a>
| |
| − | </div>
| |
| − | <div class='col-4 col-md-2 col-xl-1 sponsorLogo text-center'>
| |
| − | <a href="http://s466755640.online.de/4bl/" target="_blank2"><img class="img-fluid"
| |
| − | src="https://static.igem.org/mediawiki/2019/0/0b/T--Tuebingen--4base_logo.png"></a>
| |
| − | </div>
| |
| − | <div class='col-4 col-md-2 col-xl-1 sponsorLogo text-center'>
| |
| − | <a href="https://www.agilent.com/" target="_blank2"><img class="img-fluid"
| |
| − | src="https://static.igem.org/mediawiki/2019/e/e7/T--Tuebingen--agilent_logo.png"></a>
| |
| − | </div>
| |
| − | <div class='col-4 col-md-2 col-xl-1 sponsorLogo text-center'>
| |
| − | <a href="https://www.digitalocean.com/" target="_blank2"><img class="img-fluid"
| |
| − | src="https://static.igem.org/mediawiki/2019/6/67/T--Tuebingen--digitalocean_logo.png"></a>
| |
| − | </div>
| |
| − | <div class='col-4 col-md-2 col-xl-1 sponsorLogo text-center'>
| |
| − | <a href="https://dwk.com/" target="_blank2"><img class="img-fluid"
| |
| − | src="https://static.igem.org/mediawiki/2019/c/ce/T--Tuebingen--dwk_logo.png"></a>
| |
| − | </div>
| |
| − | <div class='col-4 col-md-2 col-xl-1 sponsorLogo text-center'>
| |
| − | <a href="https://www.eppendorf.com/" target="_blank2"><img class="img-fluid"
| |
| − | src="https://static.igem.org/mediawiki/2019/a/a6/T--Tuebingen--eppendorf_logo.png"></a>
| |
| − | </div>
| |
| − | <div class='col-4 col-md-2 col-xl-1 sponsorLogo text-center'>
| |
| − | <a href="https://www.implen.de/" target="_blank2"><img class="img-fluid"
| |
| − | src="https://static.igem.org/mediawiki/2019/b/bb/T--Tuebingen--Logo-Implen.png"></a>
| |
| − | </div>
| |
| − | <div class='col-4 col-md-2 col-xl-1 sponsorLogo text-center'>
| |
| − | <a href="https://www.jenabioscience.com/" target="_blank2"><img class="img-fluid"
| |
| − | src="https://static.igem.org/mediawiki/2019/3/31/T--Tuebingen--JBS_logo.png"></a>
| |
| − | </div>
| |
| − | <div class='col-4 col-md-2 col-xl-1 sponsorLogo text-center'>
| |
| − | <a href="https://www.gbo.com/" target="_blank2"><img class="img-fluid"
| |
| − | src="https://static.igem.org/mediawiki/2019/a/ae/T--Tuebingen--greiner_logo.png"></a>
| |
| − | </div>
| |
| − | <div class='col-4 col-md-2 col-xl-1 sponsorLogo text-center'>
| |
| − | <a href="http://www.metabion.com/" target="_blank2"><img class="img-fluid"
| |
| − | src="https://static.igem.org/mediawiki/2019/4/41/T--Tuebingen--Logo-Metabion.png"></a>
| |
| − | </div>
| |
| − | <div class='col-4 col-md-2 col-xl-1 sponsorLogo text-center'>
| |
| − | <a href="https://www.microsynth.ch/home-ch.html" target="_blank2"><img class="img-fluid"
| |
| − | src="https://static.igem.org/mediawiki/2019/1/18/T--Tuebingen--microsynth_logo.png"></a>
| |
| − | </div>
| |
| − | </div>
| |
| − |
| |
| − | <div class='row align-items-center'>
| |
| − | <div class='col-4 col-md-2 col-xl-1 sponsorLogo text-center'>
| |
| − | <a href="https://www.neb.com/" target="_blank2"><img class="img-fluid"
| |
| − | src="https://static.igem.org/mediawiki/2019/2/29/T--Tuebingen--neb_logo.png"></a>
| |
| − | </div>
| |
| − | <div class='col-4 col-md-2 col-xl-1 sponsorLogo text-center'>
| |
| − | <a href="https://www.promega.de/en/" target="_blank2"><img class="img-fluid"
| |
| − | src="https://static.igem.org/mediawiki/2019/f/f8/T--Tuebingen--promega_logo.png"></a>
| |
| − | </div>
| |
| − | <div class='col-4 col-md-2 col-xl-1 sponsorLogo text-center'>
| |
| − | <a href="https://www.qiagen.com/us/" target="_blank2"><img class="img-fluid"
| |
| − | src="https://static.igem.org/mediawiki/2019/9/92/T--Tuebingen--qiagen_logo.png"></a>
| |
| − | </div>
| |
| − | <div class='col-4 col-md-2 col-xl-1 sponsorLogo text-center'>
| |
| − | <a href="https://www.carlroth.com/en/en" target="_blank2"><img class="img-fluid"
| |
| − | src="https://static.igem.org/mediawiki/2019/6/61/T--Tuebingen--roth_logo.png"></a>
| |
| − | </div>
| |
| − | <div class='col-4 col-md-2 col-xl-1 sponsorLogo text-center'>
| |
| − | <a href="https://www.sitoolsbiotech.com/" target="_blank2"><img class="img-fluid"
| |
| − | src="https://static.igem.org/mediawiki/2019/3/31/T--Tuebingen--sibiotools_logo.png"></a>
| |
| − | </div>
| |
| − | <div class='col-4 col-md-2 col-xl-1 sponsorLogo text-center'>
| |
| − | <a href="https://www.snapgene.com/" target="_blank2"><img class="img-fluid"
| |
| − | src="https://static.igem.org/mediawiki/2019/3/3a/T--Tuebingen--snapgene_logo.png"></a>
| |
| − | </div>
| |
| − | <div class='col-4 col-md-2 col-xl-1 sponsorLogo text-center'>
| |
| − | <a href="https://www.trilinkbiotech.com/" target="_blank2"><img class="img-fluid"
| |
| − | src="https://static.igem.org/mediawiki/2019/b/b6/T--Tuebingen--trilink_logo.png"></a>
| |
| − | </div>
| |
| − | <div class='col-4 col-md-2 col-xl-1 sponsorLogo text-center'>
| |
| − | <a href="https://twistbioscience.com/" target="_blank2"><img class="img-fluid"
| |
| − | src="https://static.igem.org/mediawiki/2019/4/4c/T--Tuebingen--Logo-Twist.png"></a>
| |
| − | </div>
| |
| − | <div class='col-4 col-md-2 col-xl-1 sponsorLogo text-center'>
| |
| − | <a href="http://www.vazymebiotech.com/" target="_blank2"><img class="img-fluid"
| |
| − | src="https://static.igem.org/mediawiki/2019/b/b7/T--Tuebingen--vazyme_logo.png"></a>
| |
| − | </div>
| |
| − | <div class='col-4 col-md-2 col-xl-1 sponsorLogo text-center'>
| |
| − | <a href="https://www.biolegend.com/" target="_blank2"><img class="img-fluid"
| |
| − | src="https://static.igem.org/mediawiki/2019/c/c3/T--Tuebingen--biolegend_logo.png"></a>
| |
| − | </div>
| |
| − | <div class='col-4 col-md-2 col-xl-1 sponsorLogo text-center'>
| |
| − | <a href="https://www.zymoresearch.de/" target="_blank2"><img class="img-fluid"
| |
| − | src="https://static.igem.org/mediawiki/2019/6/6e/T--Tuebingen--zymo_logo.png"></a>
| |
| − | </div>
| |
| − | <div class='col-4 col-md-2 col-xl-1 sponsorLogo text-center'>
| |
| − | <a href="https://www.eurofins.com/" target="_blank2"><img class="img-fluid"
| |
| − | src="https://static.igem.org/mediawiki/2019/c/cd/T--Tuebingen--eurofins_logo.svg"></a>
| |
| − | </div>
| |
| − |
| |
| − | </div>
| |
| | </div> | | </div> |
| | + | <div class='col-4 col-md-2 col-xl-1 sponsorLogo text-center'> |
| | + | <a href="http://s466755640.online.de/4bl/" target="_blank2"><img class="img-fluid" |
| | + | src="https://static.igem.org/mediawiki/2019/0/0b/T--Tuebingen--4base_logo.png"></a> |
| | + | </div> |
| | + | <div class='col-4 col-md-2 col-xl-1 sponsorLogo text-center'> |
| | + | <a href="https://www.agilent.com/" target="_blank2"><img class="img-fluid" |
| | + | src="https://static.igem.org/mediawiki/2019/e/e7/T--Tuebingen--agilent_logo.png"></a> |
| | + | </div> |
| | + | <div class='col-4 col-md-2 col-xl-1 sponsorLogo text-center'> |
| | + | <a href="https://www.digitalocean.com/" target="_blank2"><img class="img-fluid" |
| | + | src="https://static.igem.org/mediawiki/2019/6/67/T--Tuebingen--digitalocean_logo.png"></a> |
| | + | </div> |
| | + | <div class='col-4 col-md-2 col-xl-1 sponsorLogo text-center'> |
| | + | <a href="https://dwk.com/" target="_blank2"><img class="img-fluid" |
| | + | src="https://static.igem.org/mediawiki/2019/c/ce/T--Tuebingen--dwk_logo.png"></a> |
| | + | </div> |
| | + | <div class='col-4 col-md-2 col-xl-1 sponsorLogo text-center'> |
| | + | <a href="https://www.eppendorf.com/" target="_blank2"><img class="img-fluid" |
| | + | src="https://static.igem.org/mediawiki/2019/a/a6/T--Tuebingen--eppendorf_logo.png"></a> |
| | + | </div> |
| | + | <div class='col-4 col-md-2 col-xl-1 sponsorLogo text-center'> |
| | + | <a href="https://www.implen.de/" target="_blank2"><img class="img-fluid" |
| | + | src="https://static.igem.org/mediawiki/2019/b/bb/T--Tuebingen--Logo-Implen.png"></a> |
| | + | </div> |
| | + | <div class='col-4 col-md-2 col-xl-1 sponsorLogo text-center'> |
| | + | <a href="https://www.jenabioscience.com/" target="_blank2"><img class="img-fluid" |
| | + | src="https://static.igem.org/mediawiki/2019/3/31/T--Tuebingen--JBS_logo.png"></a> |
| | + | </div> |
| | + | <div class='col-4 col-md-2 col-xl-1 sponsorLogo text-center'> |
| | + | <a href="https://www.gbo.com/" target="_blank2"><img class="img-fluid" |
| | + | src="https://static.igem.org/mediawiki/2019/a/ae/T--Tuebingen--greiner_logo.png"></a> |
| | + | </div> |
| | + | <div class='col-4 col-md-2 col-xl-1 sponsorLogo text-center'> |
| | + | <a href="http://www.metabion.com/" target="_blank2"><img class="img-fluid" |
| | + | src="https://static.igem.org/mediawiki/2019/4/41/T--Tuebingen--Logo-Metabion.png"></a> |
| | + | </div> |
| | + | <div class='col-4 col-md-2 col-xl-1 sponsorLogo text-center'> |
| | + | <a href="https://www.microsynth.ch/home-ch.html" target="_blank2"><img class="img-fluid" |
| | + | src="https://static.igem.org/mediawiki/2019/1/18/T--Tuebingen--microsynth_logo.png"></a> |
| | + | </div> |
| | + | </div> |
| | + | |
| | + | <div class='row align-items-center'> |
| | + | <div class='col-4 col-md-2 col-xl-1 sponsorLogo text-center'> |
| | + | <a href="https://www.neb.com/" target="_blank2"><img class="img-fluid" |
| | + | src="https://static.igem.org/mediawiki/2019/2/29/T--Tuebingen--neb_logo.png"></a> |
| | + | </div> |
| | + | <div class='col-4 col-md-2 col-xl-1 sponsorLogo text-center'> |
| | + | <a href="https://www.promega.de/en/" target="_blank2"><img class="img-fluid" |
| | + | src="https://static.igem.org/mediawiki/2019/f/f8/T--Tuebingen--promega_logo.png"></a> |
| | + | </div> |
| | + | <div class='col-4 col-md-2 col-xl-1 sponsorLogo text-center'> |
| | + | <a href="https://www.qiagen.com/us/" target="_blank2"><img class="img-fluid" |
| | + | src="https://static.igem.org/mediawiki/2019/9/92/T--Tuebingen--qiagen_logo.png"></a> |
| | + | </div> |
| | + | <div class='col-4 col-md-2 col-xl-1 sponsorLogo text-center'> |
| | + | <a href="https://www.carlroth.com/en/en" target="_blank2"><img class="img-fluid" |
| | + | src="https://static.igem.org/mediawiki/2019/6/61/T--Tuebingen--roth_logo.png"></a> |
| | + | </div> |
| | + | <div class='col-4 col-md-2 col-xl-1 sponsorLogo text-center'> |
| | + | <a href="https://www.sitoolsbiotech.com/" target="_blank2"><img class="img-fluid" |
| | + | src="https://static.igem.org/mediawiki/2019/3/31/T--Tuebingen--sibiotools_logo.png"></a> |
| | + | </div> |
| | + | <div class='col-4 col-md-2 col-xl-1 sponsorLogo text-center'> |
| | + | <a href="https://www.snapgene.com/" target="_blank2"><img class="img-fluid" |
| | + | src="https://static.igem.org/mediawiki/2019/3/3a/T--Tuebingen--snapgene_logo.png"></a> |
| | + | </div> |
| | + | <div class='col-4 col-md-2 col-xl-1 sponsorLogo text-center'> |
| | + | <a href="https://www.trilinkbiotech.com/" target="_blank2"><img class="img-fluid" |
| | + | src="https://static.igem.org/mediawiki/2019/b/b6/T--Tuebingen--trilink_logo.png"></a> |
| | + | </div> |
| | + | <div class='col-4 col-md-2 col-xl-1 sponsorLogo text-center'> |
| | + | <a href="https://twistbioscience.com/" target="_blank2"><img class="img-fluid" |
| | + | src="https://static.igem.org/mediawiki/2019/4/4c/T--Tuebingen--Logo-Twist.png"></a> |
| | + | </div> |
| | + | <div class='col-4 col-md-2 col-xl-1 sponsorLogo text-center'> |
| | + | <a href="http://www.vazymebiotech.com/" target="_blank2"><img class="img-fluid" |
| | + | src="https://static.igem.org/mediawiki/2019/b/b7/T--Tuebingen--vazyme_logo.png"></a> |
| | + | </div> |
| | + | <div class='col-4 col-md-2 col-xl-1 sponsorLogo text-center'> |
| | + | <a href="https://www.biolegend.com/" target="_blank2"><img class="img-fluid" |
| | + | src="https://static.igem.org/mediawiki/2019/c/c3/T--Tuebingen--biolegend_logo.png"></a> |
| | + | </div> |
| | + | <div class='col-4 col-md-2 col-xl-1 sponsorLogo text-center'> |
| | + | <a href="https://www.zymoresearch.de/" target="_blank2"><img class="img-fluid" |
| | + | src="https://static.igem.org/mediawiki/2019/6/6e/T--Tuebingen--zymo_logo.png"></a> |
| | + | </div> |
| | + | <div class='col-4 col-md-2 col-xl-1 sponsorLogo text-center'> |
| | + | <a href="https://www.eurofins.com/" target="_blank2"><img class="img-fluid" |
| | + | src="https://static.igem.org/mediawiki/2019/c/cd/T--Tuebingen--eurofins_logo.svg"></a> |
| | + | </div> |
| | + | |
| | + | </div> |
| | + | </div> |
| | | | |
| | | | |
| − | <script src="https://2019.igem.org/Team:Tuebingen/magni-popup-minJS?action=raw&ctype=text/javascript"></script>
| + | <script src="https://2019.igem.org/Team:Tuebingen/magni-popup-minJS?action=raw&ctype=text/javascript"></script> |
| − | <script src="https://2019.igem.org/Team:Tuebingen/popup-imagesJS?action=raw&ctype=text/javascript"></script>
| + | <script src="https://2019.igem.org/Team:Tuebingen/popup-imagesJS?action=raw&ctype=text/javascript"></script> |
| − | <div class="visualClear"></div>
| + | <div class="visualClear"></div> |
| | </html> | | </html> |