Growth Curves
Synthetic Biology was created by introducing engineering principles into the previously existing discipline of biology.
While this came with numerous advantages, one of the most important was the standardization and characterization of parts that larger biological system are built of.
Only with this toolbox of modular, well characterized parts the current achievements in companys like gingko bioworks or the teams of the iGEM competition were made possible and the biobrick standard is a great example.
Not only does this process allow for standardized parts, it also allows to critically question generally agreed on methodologies that otherwise might negatively influence either the reproducibility or performance of experiments.
However, this standardization is not fully a past achievement but an ongoing process.
We noticed during the research how to optimally grow our cyanobacteria that this process still needs alot of standardization.
This was when we decided that we want to critically question all things currently state of the art and this developed into a project in which we used expertise acquired in chemistry, physics, mathematics and biology in addition to sythetic biology.
For example, in the literature the optical density of a culture is sometimes measured with the absorption at 730 nm (SOURCE) and sometimes at 750 nm (SOURCE).
Since many labs do not have a spectrometer that is able to measure absorption at 750 nm, we decided after valuable input from James Golden to measure OD at 730 nm.
Light intensity measurements
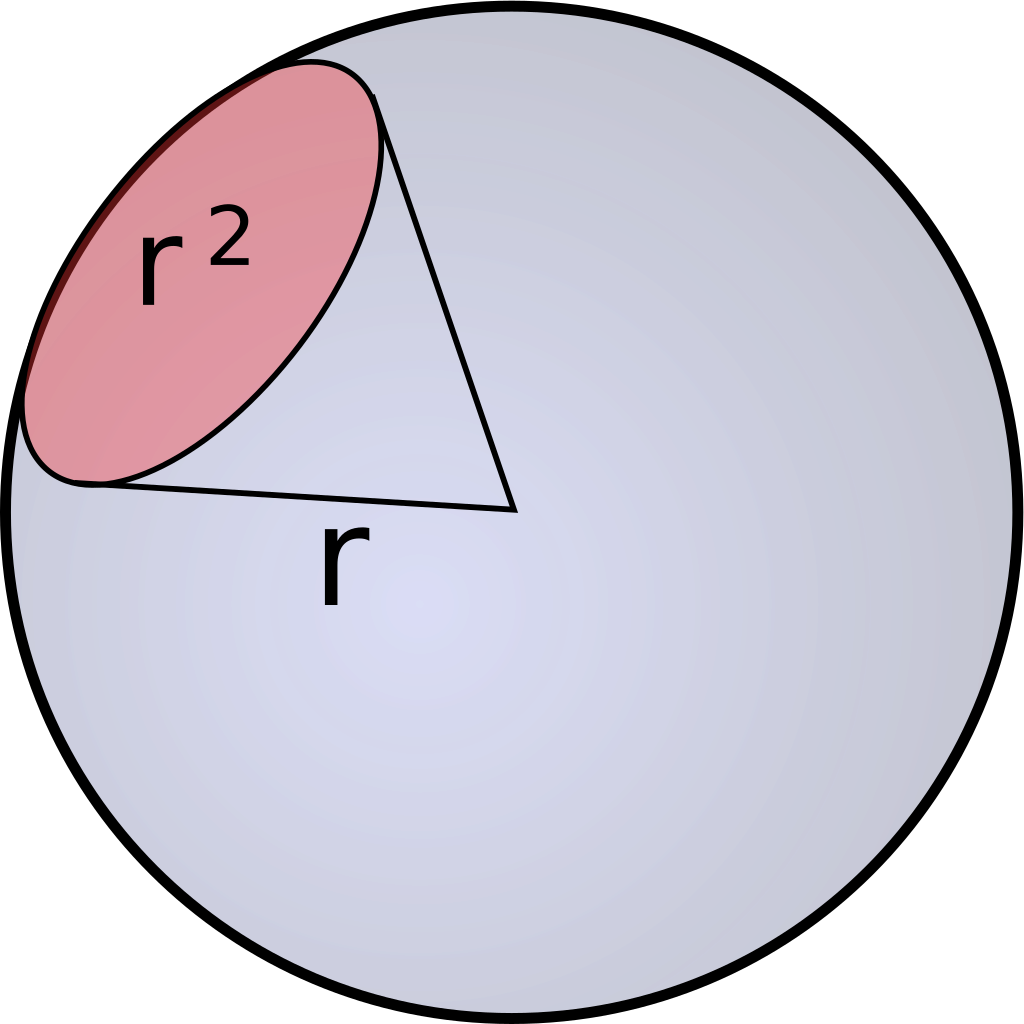
One of the first aspects that we found to be unsufficiently or contradictorily documented was the light intensity the cultures need to grow optimally. Also, the state of the art measururement for light intensity used when describing growth of cyanobacteria is Einstein, which is a non SI unit. Einstein describes the number of photons that arrive in one second at an area of one square metre, one Einstein being one mole (6.022*10**23) of photons /m2 *s. Most of the times when this unit is used in combination with photosyntetic organisms, not all photons are counted but only photosinthetically active photons with a wavelength betwenn 400 and 700 nanometers.Since Einstein is not an SI unit, there are no clear definitions how to use it which opens up possibilities for introducing errors. Due to this for example the definition of photosynthetic photons could be a point that differs between different research groups and is not specifically defined in most publications. To investigate if there is a better unit to use and if so what and how it should be used, we did an analysis of light units that we describe in the following foray:
Foray to light units

The unit Einstein, while not being an SI unit, seems to be the preferable unit for photosynthetic purposes by now, but as previously discussed it is not accurately defined. Einstein can also be expressed with only SI units as mol of photons of wavelengths between 400 and 700 nm per square meter per second. In this form, there are no insecurities about the definition and while being more voluminous to write out, we strongly believe it is much preferable. We are still using einstein as laboratory chargon to communicate more efficiently on a daily basis, however when it comes to scientific publications the SI unit version should be used at all times to circumvent communication errors.
Practical light measurements
In addition to the problems using a non SI unit introduces, the process of of measurement itself is not standardized. The light intensity could either be measured at the lamp or at the cultures, it can be measured with the cultures inside of the incubator, which yields lower light intensities due to their absorbance, or without the cultures at the place they shall be at once growing.There are two significantly different devices to measure light intensity, one with a planar sensor and one with a spherical one. Since the planar sensor has less area and also only measured light from one side it yields lower light intensities than the spherical one. Depending on the setup and how the planar sensor is used, it can also yield light intensities that are far too high (if pointed at the lamp) or too low (if pointed at e.g. the wall). The spherical measurement device gives both more reproducable and more accurate light intensities. There are empirical conversion tables to convert values measured with the planar sensor to values of the spherical measurement and vice versa, but they should be used with great caution. Again after the valuable input of James Golden we decided to use the spherical sensor to measure the light intensity at any given position.
Light model
Even with the spherical light sensor there are still some difficulties to overcome, for example where to place the cultures for a specific light intensity and that the light intensity has to be measured everytime before a growth curve. To solve both of these problems we decided to build a Light Model that models the light distribution in our incubator. With the help of this model we could enter the exact light intensity we wanted to grow the cultures at and multiple possible places were this light intesity was possible to achieve were given. The source code and the complete documentation how we designed and executed this model can be found on our Modelling page.Early Growth Curves
HERE THE EARLY GROWTH CURVES AND FACTORIAL PARAMETERS SHALL BE DISCUSSED [TORBEN/BIOLOGIST[ WE ALSO HAVE TO FIT IN A PARAGRAPH ABOUT THE wavelength we measure with With the measurement question for light solved (for us) we started to do growth curves. Many of the publications that we used as templates for our growth curves used specialized cultivation systems that were not at our disposal. With our chosen system (of erlenmeyer flasks in our incubator) there were many adjustable parameters that we stumbled upon once we wanted to do growth curves. Many of these parameters were categorial variables, but there are also some that are numerical values. We decided to do comparative growth curves for these parameters to determine which combination of parameters allows for the best possible doubling time.Flask Geometry
Fill Volume
Lid Types
Growth Curves Development
HERE THE DEVELOPMENT OF THE CURRENT METHODOLOGY FOR THE GROWTH CURVES IS EXPLAINED [BIOLOGIST]Growth Curves Model
Variables responsible for growth
As previously described, for categorial differences one can easily do growth curves with all levels of these categories (i.e. the different lids). With this it is possible to determine, at least for the chosen parameters, which level of these categories allows for the fastest growth. In reality, all parameters that play into the growth conditions of the cultures are interlinked and change when other parameters are changed. However, for some parameters the assumption that they do not change upon changing other parameters is probably a fair approximation while drastically reducing the complexity of the investigated problem. Some categorial variables (lid type, number of schikanen in the flask) are probably mostly uncorrelated with other heavily correlated parameters (light intensity, rpm, CO2, temperature) while there are others (total flask volume, filled flask volume, medium) that are more or less correlated with these parameters. While we think that the assumption of no correlation is a fair approximation for the previously mentioned categorial variables, for the fill volume of the flask we do not think it is a good approximation. This variable that as a further approximation we chose to look at as categorical has a big influence on the amount of oxygen and carbon dioxide in the flask. However, since there were already alot of variables we had to take a look at and it is heavily correlated with the CO2 percentage that we are also investigating later, we chose to fix this parameter. This would introduce a (small) error into our model, but it would reduce the complexity and the parameters of oxygen and carbondioxide in our flask can be adjusted with the concentration of carbondioxide in the incubator. For numerical parameters (light intesity, rpm, CO2 %, temperature, filled flask volume) it would also be possible to measure certain values for each variable and use the one that fits best, but there is also the possibility to model the combined effect of these parameters on the doubling time. We did the no correlation assumption of for the previously described categorial variables (lid types, flask geometry, fill volume) and developed based on biological criteria a measurement workflow for other parameters (i.e. how many precultures are used). For four other numerical parameters (temperature, carbondioxide concentration, light intensity and shaker speed) we do think that they are heavily interlinked and decided to investigate them in conjunction with each other. We used the previously established growth curve protocol and collected datapoints varying these four parameters. Due to problems with the incubator and the time constraints going with it we were not able to collect as many datapoints as we would like.Importance of a mathematical model for growth curve prediction
The data collected is displayed in the following table:| doubling_time [min] | light_intensity [µmol Photons / m2 * s 400-700 nm] | shaking speed [rpm] | CO2 [%] | temp [℃] | |
|---|---|---|---|---|---|
| 0 | 89.145 | 1500 | 130 | 5 | 41 |
| 1 | 100.014 | 1000 | 220 | 5 | 41 |
| 2 | 99.171 | 1500 | 220 | 5 | 41 |
| 3 | 96.956 | 1800 | 220 | 5 | 41 |
| 4 | 118.375 | 1800 | 130 | 5 | 41 |
| 5 | 113.305 | 1000 | 220 | 5 | 38 |
| 6 | 117.254 | 1500 | 220 | 5 | 38 |
| 7 | 122.141 | 1800 | 220 | 5 | 38 |
| 8 | 77.047 | 1000 | 220 | 3 | 41 |
| 9 | 81.442 | 1500 | 220 | 3 | 41 |
| 10 | 104.293 | 1000 | 220 | 5 | 43 |
| 11 | 96.914 | 1500 | 220 | 5 | 43 |
| 12 | 97.678 | 1800 | 220 | 5 | 43 |
| 13 | 102.040 | 1800 | 220 | 7 | 41 |
| 14 | 110.560 | 1500 | 220 | 7 | 41 |
Boundary behaviour
Something that is not part of our data are the boundaries that naturally exist for growth curves of cyanobacteria. These are partially given be the machines we are using (e.g. the maximal strength of the lamps, the maximum rpm of our shaker) and partially given by the constitution of the cyanobacteria (e.g. the maximal/minimal temperature they can grow at). With our knowledge acquired while handling this cyanobacteria, we decided on the following cutoffs:| Parameter | Value |
|---|---|
| min light [µmol Photons / m2 * s 400-700 nm] | 100 |
| max light [µmol Photons / m2 * s 400-700 nm] | 3000 |
| min rpm | 30 |
| max rpm | 260/300 |
| min temperature | 30 [℃] |
| max temperature | 50 [℃] |
| min CO2 | 1 |
| max CO2 | 10/20 |
| doubling time [min] | light intensity [µmol Photons / m2 * s 400-700 nm] | shaking speed [rpm] | CO2 [%] | temperature [℃] | |
|---|---|---|---|---|---|
| min light | 1000 | 100 | 220 | 5 | 41 |
| min rpm | 1000 | 1500 | 30 | 5 | 41 |
Modelling approach
Due to the small amount of data we were able to collect we decided to use a polynomial regression model instead of a more data demanding approach like k nearest neighbors, support vector machines or neural networks. Even with this approach, the amount of data we have at our disposal is not enough to deliver a model that we would describe as accurate within and especially not outside of our training data. Nevertheless, we think a model like this is the best way forward if we want to properly predict the doubling time and with more data a very accurate model can be built. We used a common approach to polynomial regression models in that we performed a linear regression on nonlinear functions of the data. This means that we use the previously established variables (temp, rpm, light intensity, CO2) and construct the polynomial features of this dataset. For two variables x1 and x2 and a polynomial with the degree 2 this would mean we have the following values as data : [1, x1, x2, x1*x2, x1*x1, x2*x2]. This is possible due to the fact that a linear model is not limited to a linear function but to linear parameters for the variables it builds on. The code used to build this model is shown here :Lorem ipsum dolor sit amet, consectetur adipiscing elit. Suspendisse pulvinar rutrum libero eu tincidunt. Maecenas id maximus risus, ac varius leo. Proin vulputate nulla ut sapien tincidunt varius. Donec venenatis lacinia imperdiet. Cras nulla est, varius id ornare hendrerit, viverra nec lorem. Fusce ac fermentum purus, vitae mattis ipsum. Vestibulum faucibus elit vitae vehicula tincidunt. Maecenas placerat, lorem id vulputate egestas, lectus dui faucibus urna, sed mollis nulla nulla nec ipsum. Nulla commodo viverra aliquam.

Summary and Outlook
WHAT FACTORS DID WE LOOK AT, WHERE IS FURTHER NEED FOR STANDARDIZATION, HOW CAN A STANDARDIZATION BE ACHIEVED [TORBEN/BIOLOGIST]During this investigation into how to grow UTEX2973 in the optimal way we stumbled upon many things that we thought to be insufficiently documented or standardized. We investigated how to optimally measure light intensity and thought critically about the state of the art light units. To make it possible to grow cultures at specific light intensities as well as to make a model of the light intensity in our incubator to help in the everyday life of the wetlab team. After investigating which wavelength to optimally measure the optical density of our cultures at we started to measure comparative growth curves and developed a reproducable growth curve protocol. For all parameters that had an effect on the growth curves of UTEX2973 we critically questioned if they could be approximated as independent from other parameters and decided to investigate the temperature, shaking speed, carbon dioxide concentration and light intensity in conjunction with each other. Since the investigation of four or more different dependent parameters and their effect on the growth is not exhaustively possible for humans we built an easily extendable model that uses polynomial regression to predict the doubling time of various parametercombinations. However, since measuring a single (or more) doubling time(s) is a very time demanding process, we did not manage to collect a sufficient amount of data to train a model that is able to accurately predict doubling times. In addition to "just" supplying it with more data, if we have more data more steps can be done to increase the performance of the model. In addition to a train test split and cross validation to improve the perfomance and decrease the bias of the model towards new data, LASSO regression can be used which would allow to investigate easily how high dimensional the polynome the model is utilizing has to be.


