
Can we generate DNA-free cells?
Our first objective was to generate cells without DNA. To do that, we selected 3 deoxyribonucleases produced by distinct bacteriophages, as phages are efficient and natural killers of bacteria. The first nuclease is protein A1 coming from bacteriophage T5 (Beckman et al. 1971), we named the corresponding gene nuclease_A1 (BBa_K3027000) in our project. The second nuclease is protein Gp3 produced by phage T7 (Krüger and Schroeder, 1981), we called the corresponding gene nuclease_gp3 (BBa_K3027001). Finally, we chose nuclease_yqcG (BBa_K3027002), whose product belongs to a toxin-antitoxin system. This gene is associated with phage sequences in the genome of Bacillus subtilis. Although nuclease_YqcG is annotated as a ribonuclease in databases, a published article (Elbaz and Ben-Yehuda 2015) suggested that it possesses DNase rather than RNase activity. Therefore, the 3 nucleases are believed to target mainly DNA.
DNases are toxic for cells, and unsurprisingly several iGEM teams failed to clone nucleases [1, 2, 3, 4, 5, 6]. Consequently, we needed to repress tightly the expression of the 3 nucleases. The CDS of each nuclease was cloned in the pBAD24 plasmid under the control of an arabinose-inducible promoter that is also repressed with glucose due to catabolic repression. Thanks to the addition of glucose to the medium we were successful in cloning the three nucleases in Escherichia coli.
Upon nuclease gene induction with arabinose, bacterial growth was quickly halted and bacteria lost viability. This toxicity was accompanied with reduced recovery of bacterial genomic DNA (for more detailed results, go to our web page “Demonstrate”).
Microscopic observation of cells stained with DAPI (a dye that is fluorescent upon binding to DNA) confirmed that most bacteria had lost their DNA (see an example in Figure 1B).

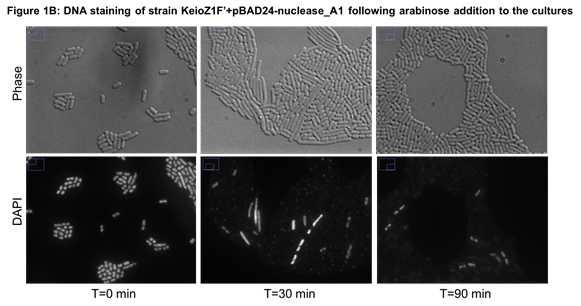 Figure 1:
Microscopy images of cells stained with DAPI to observe DNA. Bacteria
were cultured at 37°C in LB + ampicillin (100µg/mL) to logarithmic
phase before arabinose (0.2%) was added to cells. At each time point,
cells were extracted from culture medium, fixed with formaldehyde,
glutaraldehyde and mixed with DAPI. A: Control cells carrying the empty vector; B: Bacteria carrying a plasmid with nuclease_A1
Figure 1:
Microscopy images of cells stained with DAPI to observe DNA. Bacteria
were cultured at 37°C in LB + ampicillin (100µg/mL) to logarithmic
phase before arabinose (0.2%) was added to cells. At each time point,
cells were extracted from culture medium, fixed with formaldehyde,
glutaraldehyde and mixed with DAPI. A: Control cells carrying the empty vector; B: Bacteria carrying a plasmid with nuclease_A1
In conclusion, we successfully characterized the impact of 3 nucleases on E. coli cells. They degraded efficiently DNA in 30 minutes except for nuclease_YqcG which needed 90 minutes. Most importantly, DNA loss was not associated with immediate cell lysis. In some time-lapse microscopy performed with cells expressing nuclease_A1, we observed that cells maintained membrane integrity for at least 6 hours.
A predictive mathematical model to get a first answer to biological questions
For more details on the mathematical model, see our web page “Model”.
If a cell experiencing a sudden loss of DNA does not lyse immediately, we asked what happened to cell constituents (such as proteins, ribosomes or RNAs) in this kind of cells. The 2 mathematicians of the team modelized a cell without DNA. They first worked on a published model which describes with fidelity several mechanisms that happen in a cell (Weiße et al. 2015).

Figure 2: Scheme of the published mathematical model for a cell with DNA. Cellular mechanisms described in the model are in pink, cell constituents are in light blue and constants are in dark blue.
The published model describes how a cell imports nutrients, transforms them into energy (ATP) and how this energy is used by macromolecules (such as DNA polymerase, RNA polymerase or ribosomes) to perform their functions (Figure 2).
Our two mathematicians modified the model to make it describe a cell without DNA.

Figure 3: Scheme of the modified mathematical model for a cell experiencing a sudden loss in DNA. Cellular mechanisms described by our model are in pink, cell constituents are in light blue and constants are in dark blue. Note that transcription and dilution are stopped in our model.
With our unique model, we went back to ask the following question: what happens to cell constituents in a DNA-free cell? According to our model, cells constituents are still present in a cell without DNA: there are still ribosomes and the quantity of housekeeping proteins is equivalent to cells with DNA. If there are still cell constituents maybe cells without DNA can still divide. Once again, we used the mathematical model to get a first answer. To divide a cell needs to reach a critical mass (Scott et al. 2010). If a DNA-less cell has a comparable mass (i.e. number of proteins) to normal cell then it could be able to divide. From a pure proteomic point of view, a DNA-free cell could divide.
Laboratory experiments to assess some predictions from the model
As our model predicts significant results, we did the experiments. First, we extracted ribosomal RNAs (rRNA) to assess the presence of key cell constituents in cells.

Figure 4: Electrophoresis gel of genomic DNA and rRNA of KeioZ1 F’ cells producing different nucleases.
Control cells yielded a stable amount of gDNA and rRNA over time. After nuclease induction, we observed that gDNA disappeared after 30 minutes but rRNAs were not degraded, indicating that they were not cleaved by nucleases. We noticed that the bands became less intense over time (T30 vs T150 nuclease wells, Figure 4), meaning that there is a slight degradation probably due to the half-life of molecules and not the induced nucleases. As our model predicts correctly the fate of cell components, we attempted to study divisions of DNA-less cells.


Figure 5: Six hours time-lapse photography of cells carrying the empty vector (top panel) or cells producing nuclease_A1 (bottom panel). After addition of arabinose to the culture, bacteria were deposited on an agarose pad supplemented with LB, ampicillin and arabinose and covered with cover glass. Slides were observed under the microscope in a 37°C heated box. Pictures were taken every 20 minutes for 6 hours.
Control cells can divide up to 3 times on the slides (Figure 5 top). In contract, most cells producing nuclease_A1 did not finish their division , but a few cells were able to divide (bottom panel center left). As these 2 cells did not start a new division (as a cell with DNA would do) we assume they did not have DNA. Further experiments are required to confirm and quantify the percentage of DNA-free cells that can divide.
In conclusion, DNA-free cells appear to be more than just a shell of lipids: they still contain macromolecules and perform cellular functions.
“How can we repurpose a DNA-free cell?”
Inspired by our encounter with school children
Our project aimed to explore the potential of DNA-free cells. We wanted to reach different populations (e.g. biologists and non-biologists…) but also different generations. That is why we organized a meeting with children to present our project and listen to their reactions and ideas. We went to an elementary school in Viroflay (France) in June 2019 to interview children aged from 10 to 11 years old.
During our visit, we presented synthetic biology and our project using animated cartoons that we prepared beforehand. Then, we asked the children to draw what they would like to do with a cell without DNA (Figure 6).

Figure 6: Some of the children’s drawings. For more drawings and their detailed description, see our web page “Human practices”
Two thirds of the children responded with ecological bacteria, able of degrading plastic, producing energy or cleaning nature. We got inspired from their ideas and we thus explored how to give an ecological application to our DNA-less cells.
Responding to children’s request: Reprogramming our DNA-less cells into cleaning factories

Methotrexate (MTX) is an anticancer drug widely used by hospitals that is eliminated in urine and consequently rejected into wastewater. The molecule has been shown to be very toxic for fauna (Sun et al. 2009). The enzyme CPG2 (BBa_K2688003) was previously shown to biotransform MTX into glutamate and DAMPA, less toxic molecules (https://2018.igem.org/Team:GO_Paris-Saclay#/biology/cpg2).
In this project, we successfully generated DNA-free cells filled with CPG2 proteins that transformed MTX into less toxic components. DNA-less cells were able to perform metabolic degradation for at least several hours after DNA loss. Therefore, DNA-less cells can be efficiently reprogrammed into ‘cleaning factories’ for bioremediation, while reducing the risk of releasing genetically engineered microorganisms in the environment.
For more details on our DNA-free cleaning factories, go to our web page “DNA-free cell and depollution”
Limits of our DNA-free cells for genetic containment
A main limit to our DNA-free cells in genetic containment is the selection of “cheater” cells that survived and multiplied after 15 h of nuclease expression. We investigated the mechanisms of survival and found either deletions of the entire nuclease genes or the insertion of a transposase in the coding sequence (For more details about cheater cells, please visit our web page "Parts"that details the characterization of our individual biobricks). Therefore, to minimize the risks of GMO cells escaping into the environment, our lethal biobricks need to be integrated within additional strategies, such as synthetic auxotrophy or semantic containment for example (Torres, 2016).
Retracing life’s origins: from a “DNA cell” to an “RNA cell”
According to a popular theory about the evolutionary history of life, RNA
was the first molecule to carry genetic information before DNA. The theory
of an RNA world is widely accepted by scientists however no experimental
evidence supports the theory.
As a next step in repurposing our DNA-less cells, we wanted to generate an “RNA cell”, in which replicating RNA, rather than DNA, would be the support of genetic information.
We had to provide to the DNA-free cell an enzyme capable of synthesizing
RNA from an RNA template molecule, i.e. an RNA-dependent RNA polymerase.
For this, we infected the DNA-less cells with the RNA phage MS2 to bring an
RNA genome within E. coli cells. These phages encode in their genome an RNA
replicase which amplify RNA from a single strand RNA. Following infection
of DNA-free cells, we observed the production of new virions, indicating
that RNA replication and synthesis of new viral proteins took place.

Therefore, we successfully generated a DNA-less cell in which the replicating genetic information was RNA. Although ephemeral we were able to produce an RNA cell, resembling a cell that may have existed in the RNA world, before DNA evolved.
For more details on our ephemeral RNA cell, go to our web page “RNA cell”
Integrating people’s feelings about our project in the form of poems and philosophers’ thoughts
In our society, DNA means much more than just deoxyribonucleic acid. It is a ‘concept’ that society has appropriated. DNA refers to our essence, what makes sense for us and builds our identity! Modifying or removing DNA from our cells involves ethical, social and philosophical issues. We asked for people’s feedback about our project in the form of short poems. Amazed by the range of feelings displayed by all people (iGEMers, scientists and non-scientists) in response to our DNA-free cells and their potential, we created a philosophical iGAME under the form of a survey where we related each poem to the thoughts of a philosopher. This game aimed to provoke more thoughts about all the questions surrounding a DNA-free cell.
To discover many poems generated in response to our project, go to our web page “Haiku”. The results of our iGAME can be found on our web page “Human practices”.
References
[1] https://2012.igem.org/Team:Paris_Bettencourt/Human_Practice/Report
https://openwetware.org/wiki/How_safe_is_safe_enough:_towards_best_pratices_of_synthetic_biology#Preface
[2] https://2008.igem.org/Team:Waterloo
[3] https://2011.igem.org/Team:UNIST_Korea
[4] https://2013.igem.org/Team:TU-Munich/Results/KillSwitch
[5] https://2015.igem.org/Team:Hong_Kong_HKU
[6] https://2016.igem.org/Team:Exeter/Parts#section_4
Beckman, Hoffman and McCorquodale. 1971. « Pre-early proteins of bacteriophage T5: structure and function ». J Mol Biol 62: 551-564.
Elbaz
, Maya, et Sigal Ben-Yehuda. 2015. «
Following the Fate of Bacterial Cells Experiencing Sudden Chromosome
Loss
». MBio 6 (3): e00092-15. https://doi.org/10.1128/mBio.00092-15.
Krüger and Schroeder. 1981. « Bacteriophage T3 and bacteriophage T7 virus-host cell interactions ». Microbiol Rev 45 (1): 9-51.
Nikolov
, Emil N., Mariane D. Dabeva, et Todor K. Nikolov. 1983. « Turnover of Ribosomes in Regenerating Rat Liver ».
International Journal of Biochemistry 15 (10): 1255-60.
https://doi.org/10.1016/0020-711X(83)90215-X.
Scott
, M., C. W. Gunderson, E. M. Mateescu, Z. Zhang, et T. Hwa. 2010. «
Interdependence of Cell Growth and Gene Expression: Origins and
Consequences
». Science 330 (6007): 1099‑1102. https://doi.org/10.1126/science.1192588.
Sun
, S., Y. Gui, Y. Wang, L. Qian, X. Liu, Q. Jiang, et H. Song. 2009. «
Effects of Methotrexate on the Developments of Heart and Vessel in
Zebrafish
». Acta Biochimica et Biophysica Sinica 41 (1): 86-96.
https://doi.org/10.1093/abbs/gmn010.
Torres, Krüger, Csibra, Gianni and Pinheiro. 2016. «
Synthetic biology approaches to biological containment: pre-emptively
tackling potential risks.» Essays Biochem. 30 60(4):393-410. DOI:
10.1042/EBC20160013
Uehara
, T., et J. T. Park. 2008. «
Growth of Escherichia coli: Significance of Peptidoglycan
Degradation during Elongation and Septation
». Journal of Bacteriology 190 (11): 3914-22.
https://doi.org/10.1128/JB.00207-08.
Weiße
, Andrea Y., Diego A. Oyarzún, Vincent Danos, et Peter S. Swain. 2015. «
Mechanistic Links between Cellular Trade-Offs, Gene Expression, and
Growth
». Proceedings of the National Academy of Sciences 112 (9): E1038-47.
https://doi.org/10.1073/pnas.1416533112.

